Medical-Grade Monitor Manufacturer for Diagnostic & Surgical Programs
Trusted by Medical Device Manufacturers and Imaging System Partners
We support long-term OEM/ODM cooperation across diagnostic imaging, surgical display, and endoscopy system projects, helping customers reduce supply risk and maintain stable image performance across product programs.















Our display solutions are used in projects for global medical brands, device manufacturers and imaging system partners.See referenced brands & standards →
Inside Reshin Factory – Innovating Medical-Grade Monitor Manufacturing
Our factory is built for consistent output, controlled revisions, and traceability options to support long-term medical grade monitors programs.With in-house R&D, mass production capability, and strict quality control, Reshin delivers reliable OEM and ODM medical displays for global partners, ensuring long-term consistency and clinical compliance.




Core Capabilities for Medical Display Programs
From display assembly and optical design to verification and long-term supply support, Reshin helps medical display programs move forward with fewer integration surprises and more controlled project execution.

Repeatable builds and stable output for medical-grade monitor programs, supporting long-term supply of PACS diagnostic monitors and surgical imaging displays.

Support verification and calibration records to ensure consistent image performance across PACS diagnostic monitors and surgical imaging systems in clinical use.
Calibration Consistency

Offer optical bonding and front-glass options evaluated for durability, cleaning, and improved visibility in operating rooms and surgical imaging environments.
Optical Bonding Options
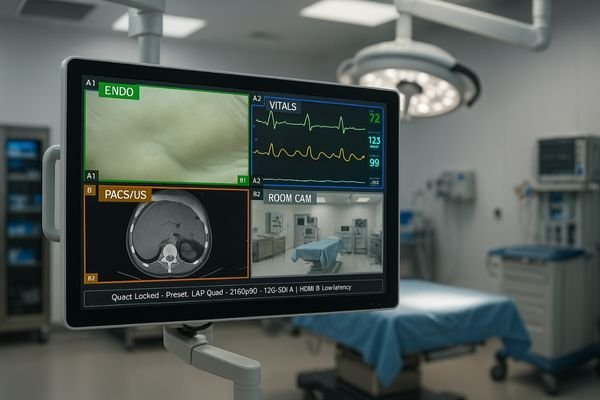
Tune signal integration for endoscopy systems and imaging devices, ensuring stable input performance for surgical and PACS monitors.
Signal Integration Support

Quality control and reliability verification processes designed to reduce field failure risks in regulated diagnostic and surgical imaging applications.
Quality Control and Reliability Verification

Lifecycle program support covering revision control, documentation, and long-term supply for OEM PACS diagnostic and surgical imaging systems.
Lifecycle Program Support
Start a Manufacturer Evaluation
Send requirements and target markets; we confirm feasibility, verification needs, and a production approach for medical grade monitors.
How a Medical Display Program Moves Forward

Step 1:Requirements Alignment
Confirm system type, interfaces, use environment, and acceptance criteria
Step 2:Feasibility Review
Evaluate customization scope, integration risks, and production feasibility.


Step 3:Sample & Validation
Provide samples for system testing, image review, and functional checks.
Step 4:Specification Freeze
Lock key parameters and verification points before ramp-up.


Step 5: Pilot Production
Validate process stability, QC checkpoints, and delivery readiness.
Step 6:Volume Supply
Support long-term supply with controlled revisions and planning continuity.

Program Manufacturing Overview
Project Support That Reduces Procurement Risk

- Specification Control
Keep key parameters and acceptance criteria aligned across shipments.
- Integration Coordination
Support interface behavior, signal stability, and system-level matching.
- Documentation Support
Prepare standard project documents and cooperate with local registration workflows.
- QC & Traceability
Provide quality checkpoints and traceability support based on project and market needs.
- Lifecycle Cooperation
Manage revisions more carefully to support long-term supply continuity.
Quality System for Medical Grade Monitors Manufacturing
Production and quality processes for medical grade monitors are controlled under ISO 13485, simplifying vendor audits and regulatory review.
CE MDR and IEC60601 compliance help secure safe, reliable performance and support faster market access.
PACS medical monitors are aligned with DIN 6868-175 to maintain diagnostic accuracy and cross-site consistency.
Conclusion: Selecting suppliers with proven certifications and hospital references reduces procurement risk and supports long-term ROI.
Get a Manufacturer Response
Share program type, size, interfaces, and target markets for medical grade monitors. We will respond with a manufacturing-ready proposal and next steps.
Yes. Reshin focuses on medical display programs for diagnostic imaging, surgical visualization, and customized OEM projects.
Yes. We support projects that require interface adjustments, front-layer changes, optical options, and program-specific coordination.
We use structured production control, verification checkpoints, and documentation support to help maintain stable output across batches.
We can cooperate on standard documentation and project coordination needed for different international market workflows.
It helps to share your system type, target size, resolution, interface requirements, target market, and project timeline.
Yes. Verification records can be prepared for medical grade monitors based on program needs and market requirements.
We verify input behavior and mode stability for medical grade monitors used with endoscopy and routing conditions.
Yes. Adjustments are supported within a controlled framework to keep medical grade monitors repeatable for long supply.
