Medical Display Solutions for Imaging, Surgical & Diagnostic Equipment
Reshin helps medical equipment manufacturers and system integrators select and integrate the right display solution for radiology, surgery, endoscopy, ultrasound, C-arm, and PACS-based systems.
- OEM / ODM manufacturing support
- Long-term model continuity
- Compliance & registration coordination
- Mechanical & I/O fit
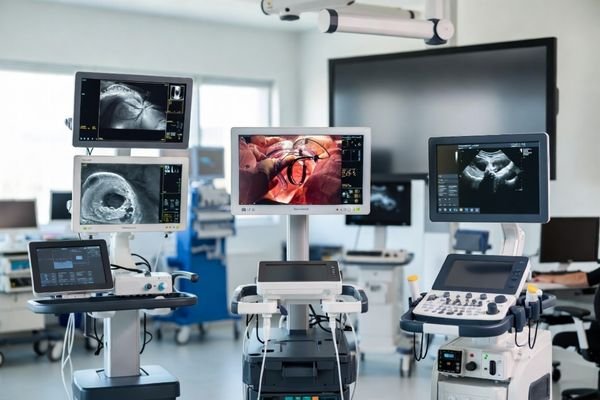
Match the display solution to the right equipment platform
Use this section to enter by system type first. Each card is written from the perspective of equipment integration, not general monitor selection.
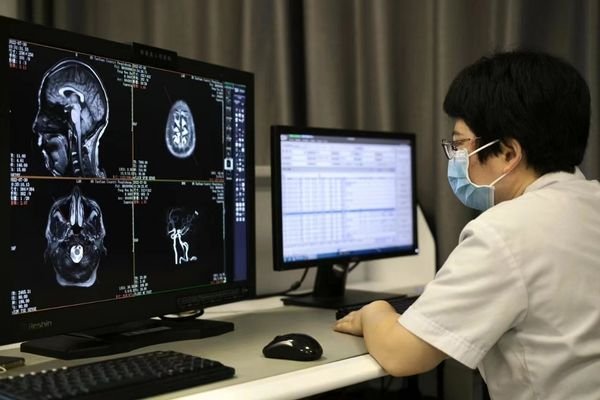
Radiology Display Solutions
For DR, CR, CT, MRI, and mammography system projects requiring grayscale consistency, workstation fit, and dependable long-term supply continuity.
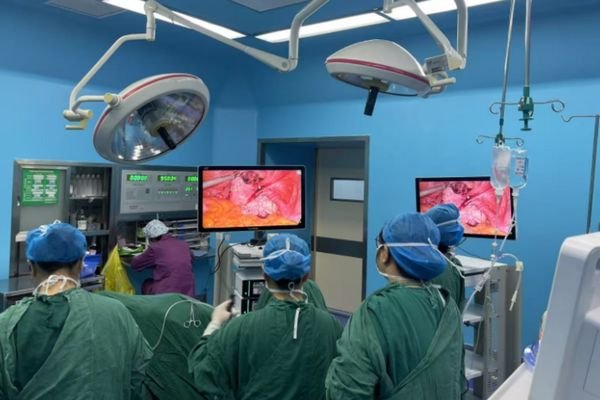
Surgical Display Solutions
For OR systems and surgical integration platforms needing cleanable surfaces, flexible mounting logic, and stable multi-display deployment.
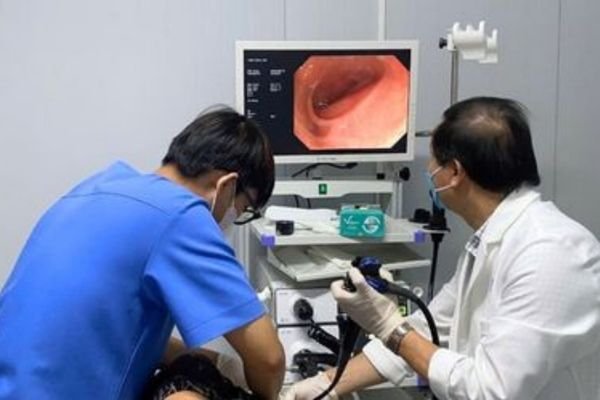
Endoscopy Display Solutions
For endoscopy towers and camera systems requiring accurate color, stable 4K video workflows, and structured integration within carts.
C-arm Imaging Solutions
For C-arm and mobile imaging equipment requiring low-latency signal handling, stable mounting structures, and reliable integrated system fit.
Ultrasound Display Solutions
For cart-based and portable ultrasound platforms needing stable grayscale rendering, wide viewing angles, and controlled long-term model continuity.
PACS Workstation Solutions
For PACS vendors and review workstations requiring reading comfort, dual-screen consistency, and dependable grayscale display performance.
Choose the support path based on project structure
Some visitors enter by equipment type. Others already know the project challenge: OEM manufacturing, compliance coordination, mechanical fit, or long-term model planning.

OEM / ODM Manufacturing
For medical equipment manufacturers and OEM brands needing controlled display manufacturing, project documentation, and pilot-to-volume supply planning.
Compliance & Registration Support
For regulated equipment programs requiring documentation coordination, engineering alignment, and project-specific compliance workflow support.
Mechanical & I/O Customization
For embedded and structure-sensitive display projects needing enclosure fit, bracket logic, and I/O layout coordination within the equipment design.
Long-Term Supply Programs
For lifecycle-controlled projects requiring model continuity, change visibility, and multi-year supply planning across regulated deployment schedules.
Project-oriented support for regulated equipment programs
This section is not about broad marketing claims. It is about the practical project conditions that matter when displays are part of a regulated medical system.
Regulated Program Continuity
Stable model planning and controlled product changes for regulated equipment programs with evaluation, validation, and deployment timelines.
Mechanical Integration Support
Support for interface mapping, mounting logic, and system-fit display selection during equipment structure and platform review.
Compliance Document Alignment
Documentation coordination for regulated equipment projects that require technical consistency across integration and registration workflows.
Lifecycle Supply Planning
Supply continuity and replacement-path visibility for programs that depend on multi-year deployment stability rather than one-time sourcing.

Review product details after selecting the right solution path
Once the equipment direction or project requirement is clear, you can move into product-level specifications to compare sizes, interfaces, resolutions, and mounting options.
Discuss Your Display Integration Project
Tell us your equipment type, screen size, interface expectations, mounting needs, and current project stage. We respond from a project and integration perspective, not from a generic monitor sales angle.
- System type, workflow, and display role within the equipmen
- Required size, interface, mounting, and housing constraints
- Project stage, evaluation timing, and supply continuity needs
- Any registration, documentation, or customization considerations
