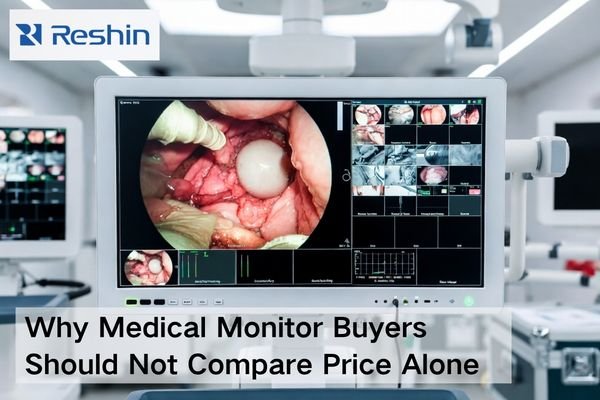
Common Compliance Mistakes When Selecting a Medical Display Supplier
Common compliance mistakes in supplier selection stem from treating certificates as a simple checkbox, ignoring documentation support, and overlooking the need for verification collaboration and change control. These mistakes occur when buyers view a display as simple hardware rather than a regulated project element requiring lifecycle management.