Selecting a long-term manufacturing partner is about repeatability and controlled change—not a single strong sample or a short-term price advantage.
The biggest risks appear after the first build: batch drift, silent substitutions, weak PCN/ECN discipline, audit gaps, and slow recovery in the field. This guide shows what to verify, what evidence to request, how to validate through EVT/DVT/PVT, and what governance to lock in so behavior stays predictable across the full OEM/ODM lifecycle.
Long-term programs succeed when stability is measurable and change is controlled. Your goal is to select a partner whose processes produce repeatable outcomes, whose documents are revision-controlled, and whose change notifications are disciplined enough to protect validated performance over time.
What "long-term manufacturing partner" really means in OEM medical displays
Manufacturing partnerships require responsibility for repeatability and controlled change management rather than delivery execution alone.
A long-term partner keeps behavior consistent across production runs while managing unavoidable change transparently. That means audit-ready quality systems, batch-to-field traceability, disciplined substitutions via PCN/ECN, and engineering collaboration that supports NPI and long lifecycle programs without drifting performance.

In OEM medical displays, “partner” means shared accountability for repeatability. The relationship must support stable outgoing tests, controlled calibration behavior (where applicable), and revision-controlled documents that remain usable for acceptance, audits, and re-validation. It also implies practical escalation: when an intermittent issue appears, the partner can trace it to batch/build, explain root cause, and prove corrective action effectiveness—rather than only swapping units.
Partnership Scope Definition and Responsibility Allocation
Long-term success starts with explicit boundaries: what is custom (ID, mechanics, sealing/cleaning, interfaces, firmware/OSD) and what must remain stable (signal behavior, test gates, calibration discipline, change rules). Confirm who owns each artifact (specs, test plans, acceptance checklists, PCN approvals), and define how decisions are documented. This prevents later disputes, reduces rework during ramp, and keeps validation aligned with program risk.
Quality System Maturity and Lifecycle Support Requirements
A long-term partner must sustain audit-ready records1, controlled revisions, and disciplined CAPA/traceability practices across the full lifecycle. Ask how they maintain documentation usability after updates, how they handle field issue investigations, and how they prove recurrence prevention. Long-term support also includes predictable change governance and continuity planning so the program remains stable as components and standards evolve.
Define the partnership scope and success metrics before you shortlist anyone
Partnership selection effectiveness increases significantly when scope definitions and success metrics are established before supplier evaluation begins.
Define scope, acceptable variation, and measurable success targets before you compare partners. Clarify what must be fixed in production, what can change with governance, and what evidence is required for acceptance. Set operational metrics such as outgoing test coverage, unit-to-unit consistency expectations, PCN lead times, continuity windows, and regional service turnaround targets.

Treat this as an internal alignment exercise as much as a supplier exercise. Engineering, quality, procurement, and program management should agree on what “pass” looks like at EVT/DVT/PVT2 and what triggers re-validation later. When scope and metrics are defined early, suppliers can be compared on evidence and capability rather than on sales language—and you reduce the risk of selecting a partner whose strengths do not match your long-term operating model.
The long-term partner scorecard: evaluation dimensions that predict stability at scale
Reliable long-term partnerships require systematic evaluation across multiple operational dimensions that determine scalability and consistency performance.
Use a scorecard that ties each dimension to evidence and verification. A stable long-term partner proves five things: mature quality systems, controlled consistency, disciplined PCN/ECN change governance, continuity/EOL planning, and responsive engineering through EVT/DVT/PVT and production ramp.

A scorecard prevents the “nice sample, weak system” trap. It forces partners to show how their processes work in production, what documents are revision-controlled, and how changes are governed. Use the table below as a working tool: you should be able to point to evidence for each category, verify it through a defined method, and identify red flags early.
| Dimension | What to verify | Evidence to request | Red flags |
|---|---|---|---|
| Quality system & traceability | ISO 13485 scope, CAPA discipline, batch-to-field linkage | Scope statement, traceability explanation, sanitized CAPA closure | No batch linkage, weak CAPA proof |
| Consistency controls | Unit-to-unit control, outgoing test gates, reject criteria | Test coverage summary, “fail criteria,” variation controls | Hand-picked samples, unclear gates |
| Change governance | PCN/ECN lead time, impact statements, approvals, regression scope | PCN template, sample impact statement, substitution rules | “No PCN for minor changes” |
| Continuity & EOL | Key-part continuity window, EOL notice, migration plan | Continuity policy, EOL policy, migration approach | Vague commitments, short notice |
| Engineering & NPI | EVT/DVT/PVT support, DFM/DFT, closure speed | NPI workflow, escalation path, drift-prevention controls | Slow closure, unclear ownership |
Quality system maturity and audit-ready traceability
Verify ISO 13485 scope, batch-level traceability, and CAPA closure discipline. Ask how a field issue maps back to batch/build and critical components, and what evidence shows corrective action prevented recurrence. A reliable partner can explain their traceability depth and provide sanitized examples, not just certificates. If they cannot investigate systematically, long-lived programs will accumulate operational risk.
Unit-to-unit consistency and outgoing test coverage
Confirm how variability is controlled across panels/components and what outgoing tests are performed. Ask what fails a unit, what gates are mandatory, and how results are monitored across production periods. The key is repeatability under normal production—not a hand-selected sample. Consistency evidence should tie to your acceptance checklist so “pass” remains stable as volumes scale.
Change control discipline (PCN/ECN)3 and regression validation
Evaluate PCN/ECN lead time, impact statement content, approval rules for critical changes, and what regression evidence is delivered. Define “critical change” clearly (panel, interface chip, firmware, calibration process, test gates) so drift cannot bypass validation. Strong partners can show templates and a consistent change-review workflow, not informal messaging.
Supply continuity, capacity planning, and EOL governance
Validate continuity windows for key parts, EOL notice periods, and migration support that preserves mechanical and integration assumptions where feasible. Ask how they manage constrained parts, how substitutions are qualified, and whether last-time-buy options exist. Capacity matters for delivery planning, but continuity and transparent governance prevent forced redesigns mid-program.
Engineering collaboration and NPI execution capability
Assess EVT/DVT/PVT support, DFM/DFT capability, test fixture planning, and issue-closure speed. Look for clear ownership, escalation paths, and documented controls that prevent sample-to-production drift during ramp. The right partner can align engineering timelines with quality gates and provide evidence-based closure, rather than informal commitments.
Evidence pack: what to request before samples and pricing decide the outcome
Minimum evidence pack requirements filter suppliers who can demonstrate sales capability but cannot sustain long-term program commitments effectively.
Request a minimum evidence pack early to verify maturity before samples dominate decisions. Focus on revision-controlled artifacts: outgoing test coverage, traceability depth, PCN/ECN practice, substitution rules, and lifecycle policies. Evidence-first evaluation reduces wasted sample cycles and prevents late-stage surprises when scaling begins.

A strong evidence pack makes partner selection defendable. It also sets expectations for how future changes will be communicated and validated. Keep the pack small but strict: require controlled revisions, clear scope, and at least one sanitized example where appropriate. If a partner cannot provide this baseline, it is unlikely they can sustain regulated, long-lived programs without friction.
Minimum Evidence Pack (baseline)
- ISO 13485 scope statement4 and quality system overview
- Outgoing test coverage summary + reject criteria (“fail gates”)
- Traceability approach + sanitized traceability record example
- Sanitized CAPA closure example (problem → root cause → corrective action → verification)
- PCN/ECN template + lead time policy
- Substitution rules + change approval workflow
- Regression validation expectations + evidence deliverable format
- Continuity policy + EOL notice period + migration approach
- Document list with revision IDs (install/clean/service/acceptance support)
Validation plan: how to prove repeatability from sample to mass production
Long-term partnership reliability requires staged validation approaches rather than single demonstration-based decision-making throughout new product introduction phases.
Prove repeatability in stages, not with a single demo. Use NPI gates to validate behavior, consistency, and production execution: EVT for basic fit, DVT for stability and multi-unit consistency, and PVT for yield, traceability execution, and packaging/shipping robustness. Freeze a baseline configuration and define re-validation triggers so change remains governable.

Staged validation forces a partner to demonstrate production reality. EVT confirms feasibility; DVT confirms repeatability under stress and across units; PVT confirms that the factory can execute consistently at scale. For each gate, agree on outputs (checklists, logs, baseline configuration) and use them as go/no-go evidence. This is how you prevent “great prototype, unstable production” outcomes.
| Gate | Goal | Sample guidance | Outputs | Go/No-Go focus |
|---|---|---|---|---|
| EVT | Prove core behavior and integration fit | Multiple units per config | Baseline tests, chain notes, issue log | Basic fit + repeatable behavior |
| DVT | Stress stability and consistency | 2–3+ units per config | Acceptance checklist, variability notes | Variability within tolerance |
| PVT | Prove production execution | Pilot build | Yield, traceability execution, packaging robustness | Repeatable production outcomes |
Multi-unit consistency testing (avoid "one perfect sample" decisions)
Test multiple units per configuration and measure results against a repeatable checklist. Include different production periods when feasible to expose drift early. Any variability should trigger root-cause explanation and corrective action evidence, not informal assurances. Consistency testing should be designed to reflect your program’s risk: what variations are acceptable, which are disqualifying, and what requires re-validation.
Real-chain stability validation5 where failures actually occur
If your deployment includes switchers, extenders, recorders, long cables, or EMI-heavy environments, validate the complete chain under realistic triggers. Test switching recovery, movement/cable stress, and stability under real room conditions. The partner should support diagnosis and mitigation guidance so issues are understood early, not discovered after launch when change is costly.
Contracts and governance: how to lock reliability into enforceable terms
Long-term manufacturing partnerships require enforceable governance frameworks that ensure accountability and performance consistency throughout extended production relationships.
Contracts should turn reliability into enforceable commitments: acceptance criteria and remedies, PCN lead times and approval rights, a clear “no silent substitution” clause, regression evidence requirements, continuity/EOL terms, and KPI reporting cadence. Governance prevents drift after launch when components change, firmware updates occur, or constraints force substitutions.

Governance should be operational, not legal theater. Your contract and operating rhythm should define what must be reported, what must be approved, what evidence must be produced, and how decisions are recorded in revision-controlled form. This is the mechanism that keeps validated behavior stable across time, production batches, and lifecycle transitions.
"No silent substitution" clause and change review workflow
Define critical changes, required notice lead time, impact statement content, and mandatory regression evidence before shipment. Establish a joint change review cadence so updates cannot bypass validation, and ensure evidence is stored in revision-controlled form. This turns “change” from a surprise into a managed process aligned with acceptance criteria and program risk.
Lifecycle continuity, EOL notice, and migration support terms
Lock a continuity window, define EOL notice period6, and require migration options that preserve mechanical/installation assumptions and integration behavior where possible. Ensure documentation updates and validation artifacts remain accessible through the lifecycle. The goal is to avoid forced redesigns and prevent last-minute transitions that break validated configurations.
Common failure patterns in long-term partnerships and how to avoid them
Systematic identification of partnership failure patterns enables proactive prevention through structured evaluation and governance approaches.
Most partnership failures follow predictable patterns: sample-to-production drift, uncontrolled substitutions, non-revision-controlled documents, unclear responsibility for intermittent issues, and service models that miss downtime tolerance. Prevent them with multi-unit testing, evidence-pack gates, enforceable PCN/ECN governance, baseline configuration freeze, and root-cause escalation—not just replacements.
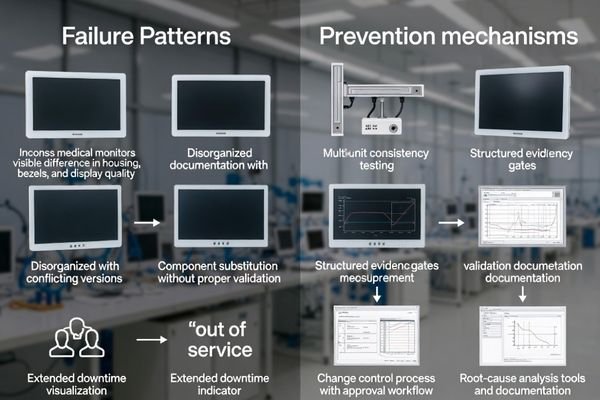
Treat prevention as a system: selection criteria, evidence requirements, validation gates, and contract governance should all reinforce each other. Then maintain it through regular reviews, KPI tracking, and change-control audits. If you only “pick a partner” but don’t govern the relationship, drift usually appears gradually—first as small inconsistencies, then as escalating operational burden.
Quick checklist for your shortlist meeting (copy-ready)
Structured evaluation checklists enable consistent and efficient supplier assessment across different evaluation teams and program requirements.
Use this shortlist checklist to keep initial calls efficient: verify ISO 13485 scope and traceability depth, confirm outgoing test coverage and unit-to-unit controls, review PCN lead time and substitution rules, validate continuity/EOL commitments, and assess EVT/DVT/PVT execution and escalation paths. The objective is to confirm what evidence exists and what must be proven before scale.

Shortlist questions to ask
- What is your ISO 13485 scope and batch-to-field traceability approach?
- What outgoing tests are performed, and what fails a unit?
- What is your PCN/ECN lead time and approval model for critical changes?
- How do you control substitutions and what regression evidence do you provide?
- What continuity window and EOL notice period can you commit to?
- How do you support EVT/DVT/PVT and prevent sample-to-production drift?
- What is the escalation path for systemic or intermittent issues?
Conclusion
Long-term medical monitor manufacturing partners should be chosen for repeatability and controlled change—not for a single exceptional sample or initial price. Evidence-first evaluation, staged validation through EVT/DVT/PVT, and enforceable governance for PCN/ECN, regression evidence, continuity, and escalation are what keep OEM programs predictable over the full lifecycle.
Reshin supports OEM/ODM display programs with validation planning, revision-controlled documentation readiness, and lifecycle governance designed to reduce drift and maintain stable behavior as requirements and components evolve.
✉️ info@reshinmonitors.com
🌐 https://reshinmonitors.com/
-
Exploring this topic will help you grasp the importance of compliance and quality assurance in the medical industry. ↩
-
Understanding EVT, DVT, and PVT is crucial for aligning engineering and procurement processes effectively. ↩
-
Understanding change control discipline is crucial for ensuring that critical changes are managed effectively, preventing issues in production. ↩
-
Exploring the significance of an ISO 13485 scope statement can help you grasp its role in quality management systems. ↩
-
Exploring Real-chain stability validation will provide insights into ensuring reliable performance in complex environments. ↩
-
Exploring the significance of EOL notice periods can help you plan for product lifecycle transitions and avoid unexpected disruptions. ↩