Buying a medical display from China for the first time is rarely just a pricing decision. For most first-time buyers, the bigger challenge is confirming that the quoted product, the approved sample, the supporting documents, and the final shipment all point to the same configuration and the same intended medical workflow. A safer first order depends on clarity, consistency, and traceable communication from the beginning.
Before placing a first order, buyers should confirm six essentials: the exact product definition, document-to-sample consistency, real workflow compatibility, written change control, reasonable limits on early customization, and the supplier’s response capability when questions or problems appear during the project.

In practice, many sourcing problems do not start with price or even with product quality. They start when the buyer and supplier are not aligned on what is actually being purchased, how it will be used, and how future changes will be handled. For first-time procurement, the goal should not be to move fastest with the fewest questions. It should be to build a procurement process that is clear enough to support stable delivery, internal approval, and long-term cooperation.
Confirm Exactly What Product You Are Buying
Many first-time procurement issues begin before the quotation stage because the buyer and supplier are not discussing the same product in the same application context. The term medical display can cover several categories, and those categories may differ significantly in intended use, image expectations, workflow requirements, and documentation needs.
Before requesting a quotation, buyers should define the actual application, signal source, screen size, resolution target, mounting method, and intended use clearly enough for the supplier to respond to the right medical display category instead of to a broad or incomplete request.
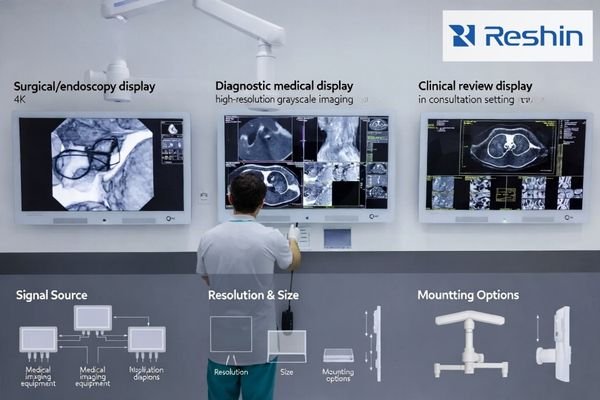
A practical first step is to describe the real use case instead of asking for a generic “medical monitor1.” Buyers should clarify whether the project involves a surgical or endoscopy display, a diagnostic medical display, a clinical review display, a consultation display, or an operating room integration display. These categories may appear similar in a quotation, but they can differ in image behavior, latency sensitivity, interface priorities, viewing conditions, mounting requirements, and applicable supporting documents. Buyers should also define the target environment, primary signal source, preferred size, whether 4K is necessary, whether multi-view is needed, how the unit will be mounted, and what market the product is intended for. This makes supplier responses more relevant and reduces the chance of receiving an unsuitable quotation or an unhelpful sample.
Application Scenario and Signal Source Definition
The real application should come first, because the workflow determines what matters most. A display used with an endoscopy processor, a surgical camera, a PACS workstation, or a digital operating room system may require different priorities for resolution, switching behavior, signal handling, viewing conditions, and response performance. Buyers should therefore define where the display will be installed, what source device it will receive signals from, and what performance expectations matter in routine use.
Use Case Parameter Specification and Supplier Alignment
A safer approach is to ask the supplier to respond against a defined use case instead of against a broad product family. In practical terms, that means confirming the application scenario, signal source type, target size, resolution expectations, multi-view requirements, mounting method, and intended market before comparing offers. This improves quotation relevance, makes sample evaluation more meaningful, and helps prevent product mismatch later in the procurement process.
Make Sure Documents, Samples, and Shipment Match
Even when a supplier provides documents and an acceptable sample, risk remains if those materials do not represent the same version that will finally be shipped. In first-time procurement, one of the most common causes of confusion is a mismatch between what was quoted, what was approved, and what actually arrives.
The approved sample, supporting documents, and final shipment should all refer to the same product configuration, because first-time medical display procurement often becomes difficult when those three elements drift apart during quotation, sample review, order confirmation, or production.

A sample is only useful if it accurately represents production. Buyers should therefore confirm that reports, available certificates, sample units, labels, accessories, and shipment records all point to the same configuration. In practice, this means checking model naming, interface configuration, panel version, accessory list, firmware identification, and label information at the sample stage, then keeping those elements aligned through order confirmation and shipment. A confirmed sample should not be treated as a simple demonstration unit. It should become the reference point for later acceptance and communication. In many first-order projects, the problem is not the absence of documents or samples. It is the lack of alignment between them. A more reliable approach is to lock the approved configuration2 in writing and use it as the baseline for future review.
Verify Real Workflow Compatibility, Not Just Interfaces
A display may have the expected connector types and still fail to perform properly in the intended medical workflow. That is why first-time buyers should not stop at interface checking. In medical projects, compatibility should be reviewed as part of the complete signal chain and real operating environment rather than as a simple connector checklist.
A matching interface alone is not enough for medical display procurement, because buyers should also verify how the display behaves in the actual workflow, including source-device behavior, timing, switching, scaling, adapter needs, cable distance, and the conditions of the real installation environment.

Interface availability does not prove workflow compatibility. Wherever possible, buyers should verify performance with the real source device, because that is often where hidden issues appear. For example, an endoscopy display should be reviewed with the actual processor or a representative source, a surgical setup should consider frame rate behavior and switching response, and a PACS-related setup should consider multi-monitor use3, cable distance, and reading workflow. The goal is not only to confirm that an image appears on screen, but to confirm that the display behaves acceptably in the intended environment. In practice, real workflow validation often reveals timing differences, switching delays, scaling behavior, adapter dependence, or installation constraints that are not obvious in a datasheet.
| Signal Source Category | Critical Compatibility Factors | Validation Requirements | Common Risk Areas | Testing Recommendations |
|---|---|---|---|---|
| Endoscopy Processors | Resolution scaling, color behavior, latency | Real processor connection testing | Proprietary signal timing, scaling mismatch | Live source testing with representative content |
| Surgical Cameras | Frame rate matching, synchronization, switching response | Multi-input workflow validation | Input lag, unstable switching behavior | Simulated operating room workflow testing |
| PACS Workstations | Multi-monitor support, viewing consistency, cable distance | Extended setup validation | Signal stability, usability in reading workflow | Test with actual workstation configuration |
| OR Integration Systems | Switching logic, signal stability, control behavior | Full system validation | Integration mismatch, unstable signal routing | End-to-end setup review before deployment |
| Teaching Systems | Large-format scaling, multi-source presentation behavior | Presentation workflow testing | Text clarity, source switching inconsistency | Validate in the intended teaching environment |
Sample Approval Is Not Enough Without Change Control
An approved sample is important, but it does not guarantee that later production will remain unchanged. In many first-order projects, buyers assume that once a sample is accepted, future shipments will automatically remain consistent. In practice, that assumption can create avoidable approval, installation, and communication risks.
An approved sample does not guarantee production consistency, so first-time buyers should define written change control rules before production begins, including what must be notified in advance, what requires reconfirmation, and how updated versions will be identified and documented.
A good sample only proves that one unit was acceptable at one point in time. It does not automatically prevent later changes in panel supply, accessories, firmware, labels, packaging, or default settings. That is why buyers should define change control4 before the first order enters production. In practical terms, the buyer and supplier should agree on which changes require advance notice, which changes require reconfirmation, how revised versions will be identified, and when supporting documents must be updated. This matters even when the revision seems small from a manufacturing perspective, because small version changes can still affect installation, internal documentation, or customer approval. A mature supplier does not prove reliability by promising that nothing will ever change. A mature supplier proves reliability by showing that changes are managed in a visible and disciplined way.
Change Notification Process Definition and Implementation
Product change notification should define what must be communicated in advance and what requires buyer reconfirmation. This may include panel-related changes, firmware updates, accessory substitutions, labeling adjustments, packaging revisions, or other modifications that can affect installation, identification, or approval. Clear written rules reduce ambiguity and give both sides a common process for handling revision events.
Production Consistency Validation and Version Management
Version management should make it easy to trace the relationship between the approved sample, the supporting documents, and the shipped units. Buyers should ask how different versions are identified, how changes are recorded, and how shipment consistency is maintained over time. This helps ensure that the first approved configuration remains meaningful after production begins.
Keep the First Order Close to the Standard Version
For first-time buyers, it is often tempting to combine supplier onboarding with multiple custom changes in the first project. However, early over-customization usually adds uncertainty at exactly the stage where clarity and control are most important. A first order is often safer when it validates the baseline first.
For a first order, buyers should stay close to the standard version whenever possible, because early over-customization can increase version complexity, document revisions, internal approval burden, and delivery uncertainty before the supplier, workflow, and product consistency have been properly validated.

The first order should usually confirm process stability rather than maximize customization. When too many custom elements are introduced at the beginning, the project becomes harder to manage because version control, document alignment, delivery timing, and approval scope all become more complicated. A lower-risk first-order strategy5 is to stay close to the standard version and delay deeper customization until the supplier, product, and workflow have already been validated. This does not mean that every adjustment must be avoided. It means the first order should focus on changes that do not create unnecessary uncertainty. The more stable the baseline version is, the easier it becomes to compare sample to shipment, manage documents, and understand what is actually being approved. Once that baseline is proven, later phases can introduce more customization through a more controlled process.
Safer first-order strategies include keeping the product definition stable, maintaining standard documentation during the initial order, validating standard production consistency before adding new variables, preserving clear version identification, and confirming the baseline product before introducing changes that may complicate internal review or expand approval scope.
Test the Supplier’s Response Capability in the First Order
A first order is not only a product test. It is also a supplier test. In many medical procurement projects, long-term cooperation quality depends less on whether the first order is perfect and more on how the supplier responds when questions, changes, or problems appear during real project execution.
The first order should also be used to evaluate how the supplier responds to technical questions, unexpected issues, and corrective-action needs, because response quality often reveals long-term cooperation quality more clearly than a smooth quotation stage or a polished sales presentation ever can.

Buyers should pay attention to how quickly the supplier answers technical questions, whether written replies define the problem clearly, whether timing commitments are explicit, whether the supplier distinguishes temporary containment from long-term corrective action, and whether after-sales communication channels6 remain active after shipment. These details often reveal more than presentation slides or early sales claims. Professional response behavior is usually visible in structure and follow-through. A capable supplier does not rely only on reassurance. It identifies the issue, explains what is known, shows what actions will be taken next, and follows through in writing. For first-time buyers, this response pattern is often a better indicator of long-term cooperation quality than a problem-free early sales stage, because real projects almost always involve uncertainty, clarification, and adjustment.
FAQ
What should I define before requesting a quotation for a medical display from China?
Before requesting a quotation, buyers should define the application scenario, signal source, target size, resolution expectations, mounting method, and intended market. This helps the supplier respond to the actual project instead of to a broad request for a general medical display.
How do I know the approved sample matches the final shipment?
Buyers should confirm that the sample, available supporting documents, labels, accessories, firmware identification, and final shipment all refer to the same configuration. For first orders, this alignment should be confirmed in writing so later approval and communication remain more controlled.
Is interface matching enough to approve a medical display?
No. A matching interface does not prove that the display will behave correctly in the real workflow. Buyers should also verify the full signal chain, especially when the project involves source timing, switching behavior, scaling, adapters, cable distance, or integrated systems.
Why is sample approval not enough by itself?
A sample only proves that one unit was acceptable at one moment. It does not prevent later changes in panels, firmware, accessories, labels, or packaging. That is why written change control is important before production begins, especially in first-order projects.
Should I heavily customize a medical display in my first order?
Usually not. For first orders, staying close to the standard version is often the lower-risk approach. Early over-customization can create version confusion, document updates, extra approval work, and added delivery uncertainty before the supplier relationship has been fully validated.
How can I evaluate a supplier during the first order?
Buyers should observe how the supplier handles questions, changes, and problems during the project. Useful signs include clear written replies, realistic timing commitments, structured problem analysis, practical corrective action, and stable after-sales communication before and after shipment.
Conclusion
Buying a medical display from China for the first time is safer when the buyer controls the process step by step. The most important checks are clear product definition, alignment between documents and the approved sample, real workflow compatibility testing, written change control, limited early customization, and close observation of supplier response quality.
A strong first order is usually not the one that moves fastest with the fewest questions. It is the one that creates a traceable and repeatable cooperation model. When buyers confirm the actual use case, lock the approved version in writing, validate the real signal chain, define change rules before production, and keep the first order close to the standard version, they reduce avoidable risk and build a more reliable foundation for future medical display procurement from China.
✉️ info@reshinmonitors.com
🌐 https://reshinmonitors.com/
-
Understanding the types of medical monitors can help you choose the right one for your specific needs. ↩
-
Understanding approved configurations is crucial for ensuring consistency and quality in production, making this resource invaluable. ↩
-
Exploring multi-monitor use challenges can help optimize PACS workstations for better performance. ↩
-
Understanding change control is crucial for buyers to ensure consistent quality and reliability in production. ↩
-
Understanding first-order strategies can help you implement effective project management practices and minimize risks. ↩
-
Exploring this link will provide insights into maintaining strong communication post-purchase, crucial for long-term supplier relationships. ↩