Understanding the distinction between medical monitor manufacturers and brand suppliers is critical for procurement decisions, integration planning, and long-term lifecycle risk management.
Medical monitor manufacturers typically control design, production, testing, traceability, and change authority, while brand suppliers typically manage sourcing, delivery, local support coordination, and SLAs. The key is not the label, but who can provide verifiable evidence and who is accountable when changes, downtime, or audits occur.

In many healthcare deployments, the wrong assumption about “who owns what” leads to preventable risk: inconsistent batches, unclear change notifications, slow escalation, or documentation gaps during audits. When you separate manufacturing authority1 from delivery/service responsibility, you can build a sourcing model that fits your operational priorities and avoids responsibility gaps over the full equipment lifecycle.
Clear definitions: manufacturer vs brand supplier
Understanding the fundamental differences between manufacturers and brand suppliers clarifies accountability and capability expectations for medical monitor sourcing decisions.
Manufacturers typically own how a monitor is designed, built, tested, and changed; brand suppliers typically own how products are selected, delivered, supported locally, and managed under service agreements.
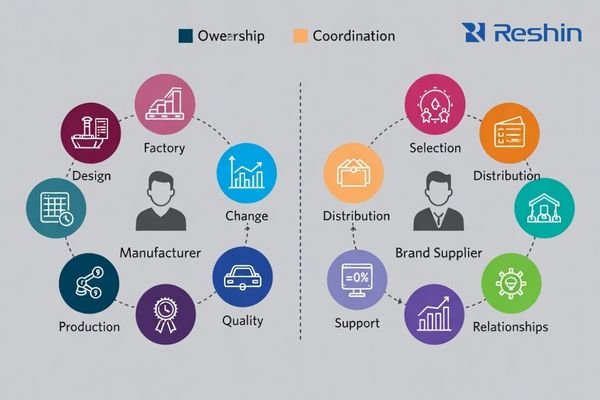
In practice, “brand supplier2” can refer to an authorized distributor, a brand owner using contract manufacturing, or a solution provider bundling products and services. Some brands are also true manufacturers, but this must be verified rather than assumed. The most reliable sourcing decisions start by confirming where engineering authority lives, who controls change notifications, and who is responsible for field outcomes.
Manufacturing Ownership and Control Capabilities
True manufacturers maintain direct control over product design processes, component sourcing strategies, production line operations, quality control systems, and engineering change management. That control typically determines fundamental behavior such as consistency across units, traceability depth, and how changes are validated and communicated over time.
Brand Supplier Service and Distribution Focus
Brand suppliers typically excel at portfolio curation, distribution logistics, project coordination, and local service management. Their value is often speed, regional coverage, and a single commercial/service window—but their technical authority depends on their contractual relationship with the underlying manufacturer.
When evaluating suppliers claiming manufacturing status, verification should focus on manufacturing authority rather than branding language: facility ownership or contracted factory relationship, quality system control, change-control authority, and direct access to engineering escalation.
Responsibilities map across the lifecycle
Supplier responsibilities vary significantly across equipment lifecycle phases, requiring clear understanding of manufacturer versus brand supplier capabilities and limitations.
Manufacturers tend to own the technical truth (build, test, traceability, change), while brand suppliers often own delivery execution (logistics, service coordination, SLA management). A lifecycle view makes the handoffs and accountability gaps visible.
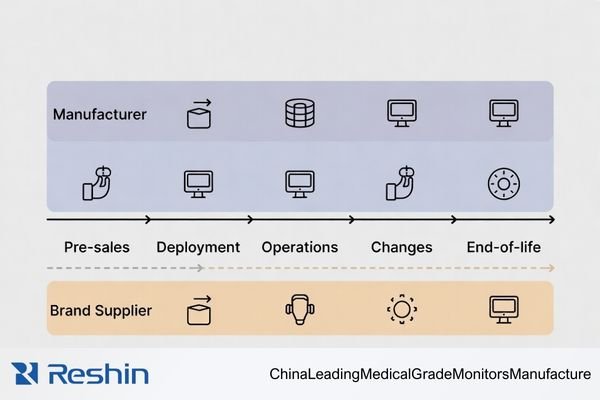
The most common sourcing failures happen when a task is “assumed” to be covered but is not assigned explicitly—especially for acceptance testing3, change notification, and escalation to factory engineering. Use the table below as a simple responsibility map for planning.
| Lifecycle phase | Manufacturer typically owns | Brand supplier typically owns | What to confirm in writing |
|---|---|---|---|
| Pre-sales / planning | Technical limits, supported behavior, change authority, evidence availability | Portfolio selection, commercial terms, pre-sales coordination | Who provides original evidence, who signs off on supported configurations |
| Deployment / acceptance | Engineering support for integration edge cases, product behavior clarification | Delivery scheduling, on-site coordination, first-line support | Acceptance criteria, escalation path, who owns acceptance failures |
| Operations / maintenance | Root-cause capability for systemic issues, controlled updates | SLA execution, RMA coordination, local spares (if offered) | RMA ownership, turnaround targets, who triggers re-validation |
| Change / lifecycle | PCN/ECN issuance, impact analysis, validation guidance | Customer communications, rollout planning | PCN lead time, approval rights, “no silent substitution” definition |
| End-of-life | EOL notice and migration options (if manufacturer-owned) | Transition coordination and local rollout | EOL notice period, migration support, continuity commitments |
A hybrid model is often the most practical: brand suppliers provide regional delivery and service coordination, while manufacturers provide evidence, change-control authority, and engineering escalation for complex or systemic issues.
Evidence and documentation: who can prove what?
Documentation authenticity and depth vary significantly between manufacturers and brand suppliers, affecting regulatory compliance and operational validation capabilities.
The key question is not who can share a certificate, but who can prove what was built, tested, and changed—using revision-controlled documents, traceability evidence, and change notifications with impact statements.
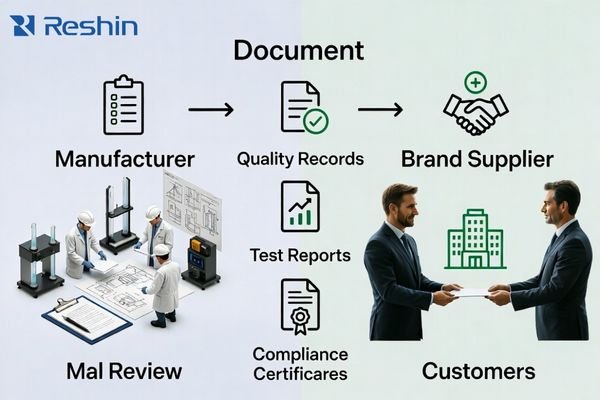
Usable documentation is operational, not decorative. You need documents you can use for acceptance, audits, maintenance, and re-validation—plus clarity on who owns revisions and change notices.
| Evidence area | Who typically owns it | What to ask for | Red flags |
|---|---|---|---|
| Quality system evidence4 | Manufacturer | Certification scope, audit approach, CAPA overview (sanitized) | No scope clarity, no corrective action evidence |
| Traceability | Manufacturer | Batch-to-field linkage explanation, example traceability record (sanitized) | No batch linkage, unclear field issue closure |
| Technical documentation | Manufacturer (original), brand supplier (distributed) | Revision history, installation/cleaning/service guides, authenticity confirmation | Mismatched revisions, “latest” not traceable |
| Change control | Manufacturer (authority), brand supplier (distribution) | PCN/ECN template, lead times, impact statement expectations | Silent substitutions, vague lead times |
| Compliance artifacts | Manufacturer (test reports), brand supplier (certificates) | Authenticity, applicable scope, supporting reports where required | Certificate-only, missing applicability to your use case |
If your procurement depends on validation, ensure you have a direct path to original evidence and revision history. When documentation is adapted by a brand supplier, confirm version alignment and how updates are synchronized with the manufacturer’s controlled revisions.
Supply continuity and change control: the biggest real-world risk
Mid-cycle product changes represent the most significant operational risk in medical monitor deployments, requiring clear understanding of control authority and notification procedures.
The biggest real-world risk is mid-cycle change: component substitutions or firmware updates that shift behavior across a fleet. Manufacturers typically control the change; brand suppliers often control how and when you hear about it.

Mid-cycle substitutions and firmware changes can break consistency by altering color behavior, interface timing, calibration characteristics, or processing latency. The solution is not “avoid change,” but govern it with clear rules and evidence.
A practical definition of “no silent substitution” should include all of the following:
- Advance notification with a defined lead time (PCN/ECN5)
- An impact statement (what changed, why, what behavior may shift)
- Approval rules for critical changes (who must sign off before shipment)
- Regression expectations (what must be re-tested, by whom, and what evidence is provided)
Manufacturing Change Control Authority
Manufacturers typically maintain authority over component substitution decisions, firmware development, and validation procedures. Confirm whether the manufacturer issues PCNs/ECNs directly, what lead time is typical, and what evidence accompanies the notice (impact analysis, test scope, version mapping).
Supply Chain Validation and Continuity Planning
Validating continuity claims requires confirming key component strategy, substitution rules, EOL policy, and how supply risks are managed. If a brand supplier is the commercial interface, clarify how continuity commitments are backed by the manufacturer and how you will be notified when constraints change.
Brand suppliers may have limited influence over upstream change decisions. To avoid communication lag, require explicit notification obligations and confirm escalation access to manufacturer engineering when a change threatens validated behavior.
Service, spares, and escalation: where downtime is won or lost
Service infrastructure and escalation capabilities determine operational reliability more than initial product specifications, requiring clear understanding of warranty ownership and technical authority.
Downtime risk is controlled by clear warranty ownership, fast RMA execution, spares strategy, and a proven escalation path to factory engineering when issues are intermittent or systemic.
Service performance depends on who owns each step: who receives the ticket, who ships replacements, who repairs, and who can authorize deeper investigation when symptoms are intermittent. Many programs fail because the commercial window and the technical authority are not connected by a clear escalation process.
A practical service verification checklist6 includes:
- Warranty ownership and RMA execution path (who processes, who replaces, who repairs)
- Repair turnaround targets and severity-based response expectations
- Spares availability strategy (local stock, central stock, lead times)
- Escalation to factory engineering for root-cause analysis and corrective action
- Evidence of closure behavior (how issues are documented and prevented from recurring)
Regional support models can reduce response time when local inventory and technicians exist, but complex issues still require a path to factory engineering. Confirm how escalations are triggered and whether the manufacturer will engage when needed.
Which one should you choose for your scenario?
Optimal supplier selection depends on specific deployment requirements, risk tolerance, and organizational capabilities rather than universal preferences or industry standards.
Choose based on your dominant risk: if delivery speed and local service are primary, a strong brand supplier can be ideal; if consistency, evidence, and change authority are primary, direct manufacturer involvement becomes critical. Many organizations use a hybrid approach to cover both.
Use this decision guide to match sourcing model to scenario:
| Scenario | Often best-fit model | What you must lock down | Typical failure mode if missed |
|---|---|---|---|
| Hospital / IDN deployments | Brand supplier + manufacturer evidence access | Warranty ownership, escalation to factory, revision-controlled documents | “Support window” without deep authority |
| OR / endoscopy integration | Manufacturer engineering involvement (direct or via supplier) | Real-chain validation, change control lead time, rapid escalation | Intermittent issues with no root-cause path |
| Diagnostic reading fleets | Manufacturer consistency + re-validation support | Calibration/QC artifacts, unit-to-unit consistency evidence, drift handling | Fleet drift and inconsistent QC behavior |
| OEM/ODM programs | Manufacturer-led (or brand + factory joint) | NPI governance, controlled change, long lifecycle commitments | Prototype-to-production drift |
| Multi-site scaling | Hybrid model | PCN/EOL governance, spares strategy, KPI-based SLA | Responsibility gaps across sites |
A hybrid approach works best when responsibilities are explicit: the brand supplier owns delivery and SLA execution, while the manufacturer provides controlled evidence, change authority, and engineering escalation.
Practical checklist: how to verify who you’re really buying from
Systematic verification procedures reveal actual supplier capabilities and accountability structures beyond marketing presentations and sales relationships.
Verification is about accountability: who owns manufacturing authority, who owns change control, who owns RMA execution, and who can provide original evidence on demand.
Use the questions below to identify the real owner of each responsibility:
- Who owns the factory relationship and quality system scope for this product?
- Who issues PCN/ECN and controls lead times and approvals?
- Who can provide original test reports, revision history, and traceability evidence?
- Who owns warranty liability and executes RMA logistics?
- Who holds spares inventory and what are the turnaround targets?
- Who can escalate to factory engineering for RCA and corrective action?
- Who defines the validated baseline configuration and re-validation triggers?
- Who owns EOL notices and migration support?
Minimum evidence pack to request before scaling:
- Certificates with scope and revision history (not certificate-only)
- Traceability explanation + example record (sanitized)
- Outgoing test coverage summary + calibration/QC approach (high level)
- PCN/ECN template + lead time policy + substitution rules
- EOL policy + notice period + continuity commitments
- RMA workflow + turnaround targets + spares policy + escalation map
Contract clauses that prevent responsibility gaps:
- PCN/ECN notice period, impact statement requirements, approval rights
- Explicit “no silent substitution” definition + regression expectations
- RMA ownership, repair/replace rules, turnaround targets, spares terms
- SLA reporting cadence + escalation timeline + corrective action expectations
- EOL notice period + migration support obligations
Verification should include direct escalation path testing (even during pilot) and confirmation that evidence can be produced in a revision-controlled form when needed for acceptance or audits.
Conclusion
Medical monitor supplier selection should optimize for evidence quality and accountability rather than labels. Manufacturers and brand suppliers often cover different operational needs: manufacturers anchor consistency, traceability, and controlled change, while brand suppliers often excel at delivery coordination and local service execution.
Our experience at Reshin shows that the most reliable approach is frequently a hybrid model with explicit responsibilities: brand suppliers manage delivery and service windows, while manufacturers provide original evidence, change-control authority, and engineering escalation. When organizations verify ownership, lock change governance, and define escalation paths in advance, downtime risk and lifecycle surprises decrease.
✉️ info@reshinmonitors.com
🌐 https://reshinmonitors.com/
-
Understanding manufacturing authority is crucial for effective healthcare operations and risk management. ↩
-
Understanding the role of brand suppliers can enhance your sourcing strategy and supplier evaluation process. ↩
-
Understanding acceptance testing is crucial for ensuring project deliverables meet requirements and expectations. ↩
-
Understanding quality system evidence is crucial for ensuring compliance and effective audits in manufacturing processes. ↩
-
Exploring PCN and ECN will provide insights into how manufacturers communicate changes and maintain product integrity. ↩
-
A comprehensive service verification checklist can enhance operational efficiency and ensure all critical aspects are covered. ↩