Finding a reliable medical display supplier requires evidence-based evaluation of supply continuity, quality consistency, documentation readiness, integration stability, and service support—not just a review of product specifications.
A reliable medical display supplier proves five things: stable supply, consistent quality across batches, usable compliance documentation, predictable performance in real clinical setups, and responsive service. The safest way to choose is to define measurable targets, request verifiable evidence, validate with multiple units in real conditions, and lock expectations in contracts and SLAs.

Supplier reliability1 directly affects rollout timelines, downtime risk, and the long-term effort required to keep a fleet stable. A structured approach reduces surprises by turning “reliable” into measurable criteria, then confirming those criteria through documents, testing, and enforceable commercial terms.
What does "reliable" mean for a medical display supplier?
Reliability encompasses multiple operational dimensions that ensure consistent supplier performance throughout extended partnership relationships.
Reliability means consistent supply, consistent product behavior, audit-ready documentation, stable integration performance, and predictable support response—measured with clear metrics rather than subjective impressions.
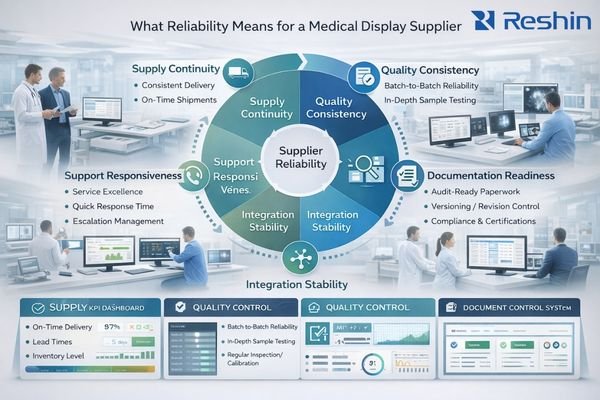
Reliability should be defined in the context of your deployment scenario. A surgical cart deployment may prioritize signal stability and fast service turnaround, while a radiology fleet may prioritize unit-to-unit consistency and repeatable QC workflows. Start by deciding which dimensions are “must not fail,” then set measurable targets so performance can be reviewed objectively.
Supply and Quality Reliability Metrics
Supply reliability can be tracked with on-time delivery rate, lead-time consistency, and clear communication about risk and end-of-life timelines. Depending on program criticality, organizations often set targets such as high on-time delivery performance and defined lead-time windows, supported by a supplier’s demonstrated inventory and continuity strategy.
Quality reliability is about batch consistency and field outcomes. Useful measures include RMA rate, unit-to-unit variation within agreed tolerances, and repeatable calibration behavior for fleets. Pair metrics with process evidence: outgoing test coverage, traceability depth, and how corrective actions are documented and closed.
To make reliability operational, define a small dashboard of supplier KPIs2 (OTD, RMA, MTTR, PCN lead time, documentation completeness) and review them on a fixed cadence. This turns reliability into governance rather than a one-time selection decision.
Which supplier type should you source from, and what risks come with each?
Different supplier channel types offer distinct advantages and risk profiles requiring careful evaluation for specific procurement scenarios.
Common sourcing paths include manufacturer-direct, authorized distributors, and systems integrators—each can be reliable, but only if authorization, warranty ownership, escalation, local service, and spares access are clearly verified.
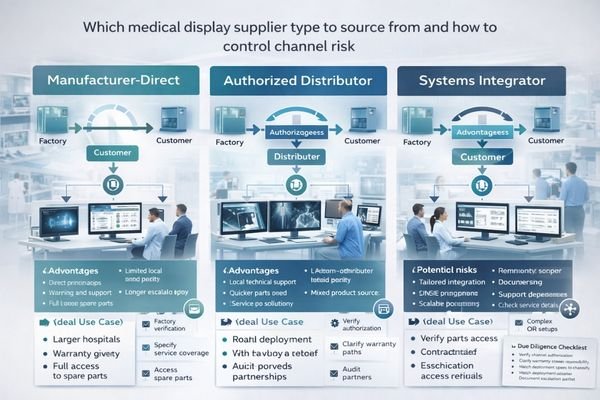
Channel choice should be based on the operating model you can support. Manufacturer-direct can be ideal for standardized fleets, long lifecycle planning, and engineering escalation. Authorized distributors can be ideal when local inventory, rapid logistics, and regional service matter. Systems integrators3 can be ideal when you need installation, configuration, and end-to-end accountability for a room build.
Use the checklist below to control channel risk:
- Verify authorization scope and territory in writing (especially for distributors)
- Define warranty responsibility and who owns RMA execution
- Confirm escalation path to manufacturer engineering for complex issues
- Confirm local service coverage, spare parts access, and turnaround expectations
What evidence should you request before you trust a supplier?
Comprehensive evidence validation reveals actual supplier capabilities beyond marketing presentations and sales commitments.
Before committing, request a minimum evidence pack covering traceability, test coverage, change control, compliance documentation, supply continuity, and service processes—then verify that the documents are current, revision-controlled, and usable for acceptance and re-validation.

Use the table as a practical tool: it lists what to request, why it matters, and what “red flags” look like.
| Evidence category | What to request | Why it matters | Red flags |
|---|---|---|---|
| Quality systems4 | Traceability approach, outgoing test coverage summary, CAPA overview | Predictable quality and field issue closure | No batch linkage, weak corrective action evidence |
| Compliance documentation | Current certificates, revision history, install/cleaning/service docs | Audit and acceptance readiness | Expired/mismatched scope, missing revision control |
| Change control | PCN/ECN template, lead-time policy, substitution rules, regression approach | Prevents silent drift and surprises | Silent substitutions, unclear approvals/lead times |
| Supply continuity | Continuity statement, key component risk approach, EOL policy/notice | Protects production and long deployments | Vague continuity, no EOL plan, single-source risk |
| Service infrastructure | RMA workflow, escalation path, spares policy, turnaround targets | Predictable recovery from issues | No spares plan, unclear escalation, vague turnaround |
Keep the evidence pack “minimum” and enforceable. If a supplier cannot provide revision-controlled documents and clear process definitions, reliability will be difficult to maintain even if initial samples look good.
How do you validate performance and consistency with samples and pilots?
Sample testing and pilot programs provide essential validation of supplier claims under realistic deployment conditions.
Validate reliability by testing multiple units per configuration and running a short pilot in real conditions that stress the full setup—signal chain behavior, switching, mounting/cable stress, and cleaning routines.

A reliable sample plan tests for variability, not just “does it work.” Use at least 2–3 units per configuration to check unit-to-unit consistency, and expand sampling for high-risk environments. Where possible, include units from different production periods or batches to reveal consistency control.
Comprehensive Sample Testing Strategy
Test multiple units under the same setup and acceptance checklist, focusing on repeatability: stable signal behavior, predictable interface handling, and consistent visual behavior where it matters for your use case. Record outcomes in a simple scorecard so the decision is evidence-based and comparable across suppliers.
Real-World Environment Validation
Design pilots to mirror real deployment triggers: full signal chain, typical switching events, routine cart movement or mounting adjustments, and cleaning steps that may stress connectors and surfaces over time. The goal is not to create a perfect lab environment, but to expose failure modes that appear only under operational stress.
Pilot outputs should be concise and reusable: an acceptance checklist5 with pass/fail criteria, an issues log with closure behavior and timelines, and a baseline configuration that defines what was validated and what triggers re-validation.
How do you structure contracts and SLAs to keep supply reliable long-term?
Strategic contract structure and service level agreements ensure predictable supplier performance throughout extended partnership relationships.
Strong contracts translate “reliability” into enforceable terms: acceptance criteria, warranty and RMA rules, spares and turnaround targets, change-control obligations (PCN lead time and no silent substitutions), and lifecycle commitments including EOL notice and migration support.
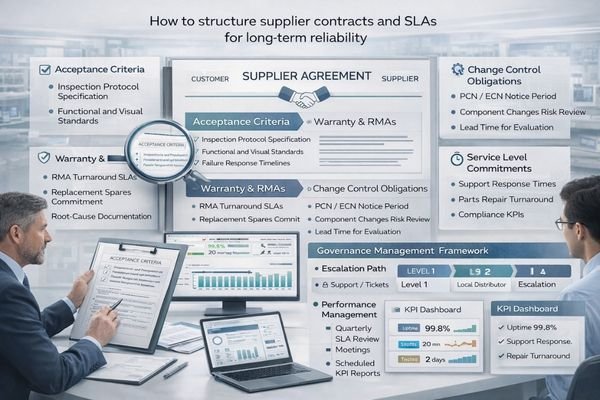
To keep contracts actionable, focus on four pillars:
- Acceptance and warranty: acceptance criteria, warranty scope, and RMA workflow
- Change control: PCN lead time, approval rights for critical changes, and regression expectations
- Lifecycle: supply continuity window, EOL notice period, and migration support approach
- Governance: escalation path, regular reviews (QBR), KPI reporting, and continuous improvement actions
You can add optional risk clauses (financial stability checks, backup sourcing expectations, IP protections) based on program sensitivity, but the core reliability outcome comes from measurable SLAs and disciplined change governance.
FAQ
Should I buy from the manufacturer directly or from an authorized distributor?
Manufacturer-direct can suit large fleets and lifecycle commitments; distributors can provide local stock and faster support. Choose the channel that proves authorization, defines warranty ownership, and has a clear escalation path to the factory.
What evidence proves a supplier is reliable beyond sales promises?
Request traceability and change-control proof: batch tracking, outgoing test coverage, CAPA handling, and PCN/ECN process with lead times. Look for revision history and real issue-handling examples.
How many sample units should I test before placing a larger order?
Test multiple units—typically at least 2–3 per configuration—to check unit-to-unit variation. For high-risk environments, expand samples and run a short pilot under real triggers.
What contract terms matter most for long-term supply reliability?
Lock acceptance criteria, RMA/turnaround targets, spare parts terms, and a "no silent substitution" clause with defined PCN lead time. Include lifecycle terms like supply continuity window, EOL notice period, and migration support.
How can I prevent mid-cycle changes from breaking consistency across my fleet?
Require written change notifications, approval rights for critical changes, and regression validation tied to your acceptance checklist. Maintain a validated baseline configuration and re-validate when changes affect behavior.
What are the biggest red flags that a supplier may not be reliable?
Unclear authorization, vague warranty ownership, missing revision-controlled documents, inconsistent lead times, and reluctance to share traceability/change-control details. Another red flag is strong samples but inconsistent batch deliveries.
Conclusion
Finding a reliable medical display supplier is a repeatable process: define reliability with measurable targets, select the right sourcing channel, demand a minimum evidence pack, validate with multi-unit testing and a realistic pilot, and lock expectations into SLAs and change-control terms. This approach reduces procurement risk and supports predictable operations across clinical deployments.
Our experience at Reshin shows that reliability is proven through revision-controlled documentation, repeatable acceptance, and predictable support response over time—not through sales claims or single-unit impressions. When organizations govern suppliers with clear metrics and disciplined change management, fleet stability improves and lifecycle surprises decrease.
✉️ info@reshinmonitors.com
🌐 https://reshinmonitors.com/
-
Understanding supplier reliability is crucial for minimizing risks and ensuring timely project rollouts. ↩
-
Exploring key supplier KPIs can help you establish effective metrics for monitoring and improving supplier performance. ↩
-
This resource will help you understand the critical role of Systems integrators in ensuring successful installations and configurations. ↩
-
Understanding traceability in quality systems is crucial for ensuring predictable quality and effective issue resolution. ↩
-
Understanding acceptance checklists is crucial for ensuring that testing meets all necessary criteria and standards. ↩