Medical Displays Designed for Long-Term Medical Equipment Lifecycles
For medical equipment manufacturers and project owners, regulated programs require predictable availability, stable model consistency, and medical display lifecycle management—so validation stays intact while deployments remain reliable over time.
- Long-term supply medical display planning aligned to regulated project timelines and production waves
- Stable model medical monitor supplier strategy to reduce qualification churn and deployment disruption
- Controlled change & documentation to support validation, traceability, and audit readiness
- Medical equipment display supply continuity for multi-site rollouts and long-term service replacement
Built for programs that require a medical monitor long lifecycle approach and consistent configurations.
Why Long-Term Supply Matters in Medical Equipment Projects
Medical equipment projects often span years—from verification and regulatory approval to production, multi-site deployment, and service replacement cycles. Unplanned display changes can trigger re-validation, documentation updates, and schedule risk.
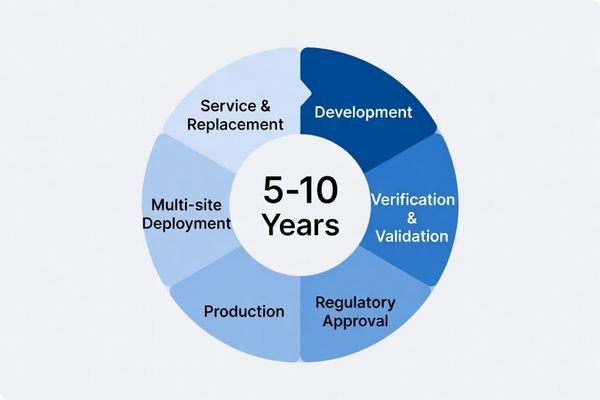
What can go wrong without lifecycle control
- Model changes impact validated configuration and system documentation
- Inconsistent batch behavior increases QA review and acceptance friction
- Supply interruptions delay production waves and multi-site deployments
- Field replacement becomes difficult when compatible models are unavailable
Supply
Program-aligned long-term availability planning
Model
Stable configurations to reduce qualification churn
Change
Controlled revisions with clear documentation
Service
Replacement strategy across extended deployments
Typical Medical Equipment Projects Requiring Long-Term Supply
These scenarios commonly require long-term supply medical display planning, controlled revisions, and predictable production availability.
Why these projects choose a long lifecycle approach
In regulated environments, selecting a stable model medical monitor supplier helps maintain consistent performance, preserve validated configurations, and reduce disruption to qualification and deployment. This supports medical display lifecycle management and keeps multi-site programs aligned.
- Supply continuity planning to support production waves and long-term deployment
- Controlled revision history aligned with documentation and regulatory expectations
- Predictable procurement and manufacturing schedules with fewer unplanned substitutions
Regulated Imaging Platforms
Stable displays for systems with long approval cycles and strict traceability requirements across product generations.
- DR / CT / MRI acquisition and review workflows
- Consistency across manufacturing waves and sites
Surgical & Endoscopy Systems
Consistent performance across production batches to support OR integration, standardized rollouts, and service continuity.
- Endoscopy carts and operating-room display chains
- Standardized configurations across hospitals/regions
Diagnostic Monitoring Programs
Long deployments where device stability and documentation consistency help reduce re-qualification and training overhead.
- Multi-year installations with fixed requirements
- Clear revision history for QA and procurement
OEM Locked Configurations
Programs that require repeatable displays over years benefit from controlled BOM, predictable availability, and change notices.
- Multi-year supply contracts and service replacement planning
- Stable specifications aligned to regulated documentation
Key Challenges in Long-Term Display Supply
Long lifecycle programs need stability across models, batches, and availability windows to reduce validation and deployment risk.
Unexpected Model Changes
When models change without control, teams may face re-validation, updated documentation packages, and delayed production ramps.
- Qualification scope expands unexpectedly
- Deployment schedule becomes harder to maintain
Batch-to-Batch Variability
Variability across production batches can increase QA checks and complicate acceptance—especially when systems ship globally.
- More QA effort and longer acceptance cycles
- Higher risk of inconsistent user experience
Supply Disruptions Over Time
Component EOL and substitutions can break medical equipment display supply continuity, impacting both production and service.
- Procurement disruption and line changes
- Service replacement compatibility risk
How Reshin Ensures Long-Term Supply & Model Consistency
This approach supports medical display lifecycle management and reduces program risk by keeping configurations stable, changes controlled, and availability predictable across the lifecycle.
Baseline Definition
Lock key specifications and interfaces for your platform release, so validation remains stable and repeatable.
Configuration Control
Maintain a stable BOM and a controlled revision plan aligned to documentation and regulated project expectations.
Availability Planning
Forecast supply windows and manage component lifecycle transitions to reduce procurement disruptions.
Consistency Monitoring
Batch consistency checks help protect validated performance and reduce QA friction across production waves.
What You Get
- Long-term medical display supply programs aligned to regulated timelines
- Stable model definition and specification locking to reduce qualification churn
- Traceability support for regulated submissions, audits, and revision history
- Replacement/service planning across extended deployments and regional rollouts
- Controlled component changes with advance communication and documentation alignment

Typical Long-Term Supply Scenarios
A common need is medical equipment display supply continuity across manufacturing waves, multi-site installations, and service replacement cycles.
| Scenario | Risk if Uncontrolled | Risk-Control Approach |
|---|---|---|
| 5–10 year availability requirement | Re-qualification and procurement disruption when models change or components reach end-of-life. | Roadmap planning + controlled revisions aligned to documentation and validation scope. |
| Multi-site deployments | Mixed models lead to inconsistent user experience and complicate training, service, and acceptance. | Stable model rollout + batch consistency checks across production waves and regions. |
| Service & replacement over time | Field replacements break validated configuration and create documentation drift across installed base. | Service continuity planning + compatible substitutions (controlled and documented). |
| OEM platform refresh cycles | Unplanned changes ripple into documents, approvals, and manufacturing schedules. | Change notices aligned to documentation and regulatory needs with predictable transition planning. |
Recommended Medical Display Characteristics for Long-Term Projects
Selecting displays designed for lifecycle stability helps reduce re-qualification effort and supports predictable performance across production batches and service replacement.
Recommended characteristics
- Displays designed for long lifecycle availability and stable sourcing
- Stable specifications suitable for regulated environments and documentation
- Predictable performance across production batches
- Interfaces and form factor supporting platform standardization
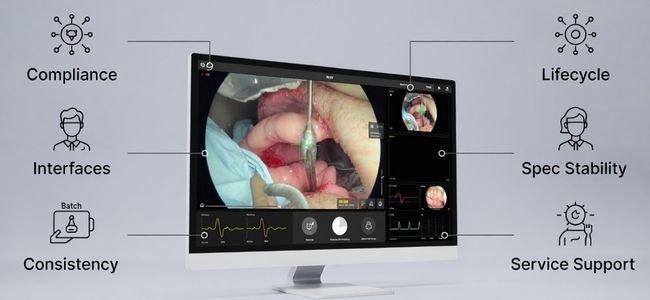
Looking for a Long-Term Display Partner for Medical Equipment Projects?
If you are developing or manufacturing medical equipment that requires long-term supply stability and controlled display lifecycles, Reshin supports regulated projects with predictable availability, stable model consistency, and documented change management.
- Long-term supply medical display planning aligned to approval cycles, production waves, and service windows
- Stable model consistency to reduce qualification churn and keep multi-site deployments standardized
- Change-control discipline with advance notice and traceability for regulated documentation
- Medical equipment display supply continuity to support service replacement and installed-base stability
