Selecting the right medical display manufacturer for OEM projects requires a structured assessment of engineering capability, quality discipline, and long-term support—not just a comparison of display specifications.
Top medical display manufacturers for OEM projects stand out through controlled customization, audit-ready quality systems, disciplined change control, supply continuity, and responsive engineering support. The goal is not to find a universal “best,” but to shortlist the best-fit manufacturer profile for your OEM scope and validate it through repeatable evidence and a real-world pilot.
In OEM programs, manufacturer selection shapes outcomes long after the first units ship. The right partner helps you maintain consistency across production runs, document decisions for audits, and manage unavoidable changes without drifting behavior in the field. A systematic evaluation1 reduces risk across NPI, scaling, and lifecycle support—especially when your product will be deployed across many sites, rooms, and years.
What "OEM-ready" means for medical displays
OEM-ready manufacturers provide comprehensive customization and lifecycle support beyond standard product offerings for integrated medical device development.
OEM-ready means the manufacturer can support customization, supply continuity, controlled change management, and consistent behavior at scale—so your product remains stable from prototype through production and throughout its lifecycle.
From an engineering perspective, OEM risk2 usually comes from what happens after the first prototype: uncontrolled substitutions, inconsistent calibration behavior, documentation gaps, or unclear lifecycle policies. A strong OEM-ready partner can collaborate during development while also proving that production controls, traceability, and change governance are mature enough to protect your product in the field.
OEM Partnership Requirements vs Standard Purchasing
OEM partnerships require more than purchasing transactions. The manufacturer must support iterative engineering work, align on test and acceptance criteria, and keep production behavior consistent as volumes grow. This includes predictable communication on changes and a practical mechanism to verify that updated builds still match validated performance.
Comprehensive Scope of OEM Customization Capabilities
Typical OEM scope may include industrial design updates, mechanical modifications, sealing and cleaning compatibility considerations, interface and signal-behavior alignment, firmware/OSD adjustments, labeling and traceability rules, and packaging optimized for distribution and integration workflows. The key is not the breadth of options, but whether customization is delivered under controlled documentation and repeatable validation.
The evaluation criteria that matter most for OEM projects
Critical evaluation criteria focus on operational capabilities that enable successful long-term OEM partnerships beyond basic product performance.
The strongest OEM manufacturers prove five things with evidence: disciplined quality systems and traceability, audit-ready compliance documentation, engineering collaboration through EVT/DVT/PVT, supply-chain consistency controls, and lifecycle/service readiness that scales with your program.
Based on typical OEM workflows, it helps to evaluate each category using what you can verify—not what is promised. Use the table below as a practical scoring tool by requesting the specific evidence that demonstrates maturity.
| Evaluation category | What to verify | Evidence to request | Red flags |
|---|---|---|---|
| Quality systems | Traceability, test discipline, CAPA maturity | Sample traceability record, test coverage overview, CAPA example (sanitized) | No batch linkage, inconsistent test gates |
| Change control | How firmware/material changes are reviewed and communicated | PCN/ECN template, lead time policy, regression approach summary | Silent substitutions, unclear approvals |
| Engineering collaboration | NPI execution through EVT/DVT/PVT | NPI timeline example, DFM/DFT approach, escalation path | Slow closure, unclear ownership |
| Supply continuity3 | Key component strategy and continuity commitments | Key component list policy, substitution rules, continuity statement | Single-source risk without plan |
| Performance stability | Predictable behavior in real chains | Representative test results, stability scenarios covered | Dropouts, unstable timing, inconsistent processing |
| Lifecycle/service | Spares, repair turnaround, EOL policy | Spares policy, turnaround targets, EOL notice policy | No spares plan, vague EOL migration |
Supply-chain robustness is not only about having suppliers—it is about preventing unplanned variability. A manufacturer should be able to explain how substitutions are controlled and how they ensure unit-to-unit behavior remains stable over time. Likewise, performance stability should be discussed in the context of real integration environments rather than ideal lab conditions.
A practical shortlist process: from RFI to mass production
Systematic shortlisting processes reduce OEM risk through structured evaluation stages that validate manufacturer capabilities progressively.
A practical OEM shortlist process moves through RFI → RFQ → engineering samples → pilot build → mass production governance, with clear go/no-go criteria and concrete deliverables at each gate.
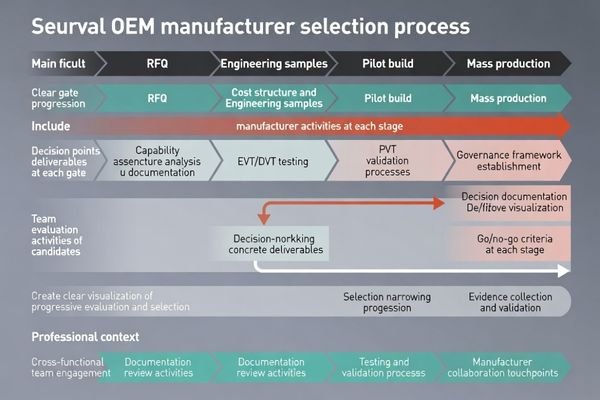
In complex OEM programs, the fastest way to reduce risk is to make each phase produce a decision artifact4. The following checklist shows what each stage should deliver before you proceed.
- RFI: confirm capability boundaries, quality system maturity, traceability approach, and documentation scope
- RFQ: compare costs by true drivers (customization, test, certification support, service), not only unit price
- Engineering samples (EVT/DVT): validate behavior in representative setups and verify repeatability across multiple units
- Pilot build (PVT): validate yield, batch consistency, traceability execution, and packaging/shipping robustness
- Mass production: lock PCN/ECN rules, version control, and regression expectations to prevent drift during lifecycle
A key principle is to validate consistency early. Even if the first sample looks excellent, OEM success depends on whether the manufacturer can keep output stable when volumes scale and when supply-chain realities force controlled changes.
Top manufacturers by OEM-fit categories (not strict ranking)
Manufacturer categories reflect different strengths and optimal use cases rather than hierarchical rankings for OEM project success.
For OEM projects, “top” manufacturers are best understood as best-fit categories: some excel at design-to-manufacture customization, some at audit-ready compliance discipline, some at high-volume continuity, and others at application-specific stability for surgical or diagnostic workflows.
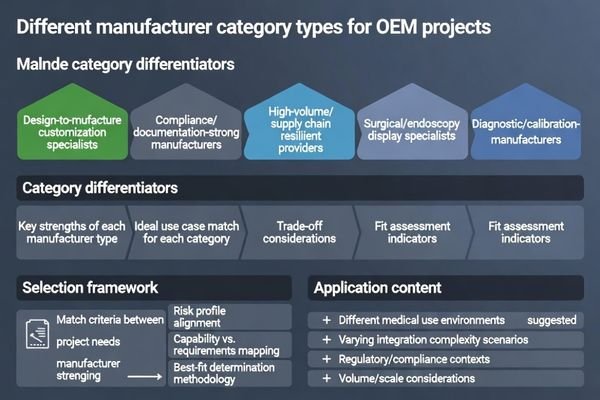
To shortlist effectively, match the category to your program risk. A customization-heavy project often needs deep engineering bandwidth, while a regulated deployment may prioritize documentation discipline and stable revisions. High-volume programs typically prioritize continuity and consistent behavior across production runs.
Customization-Focused5 and High-Volume Categories
Full-service OEM/ODM partners are best for heavy customization and fast NPI when you need design iteration and manufacturing alignment under one roof. The trade-off is that you must validate their change control rigor and proof of consistency, because customization without disciplined governance can increase drift risk.
High-volume, supply-chain resilient manufacturers are best for stable long-term supply with predictable lead times at scale. The trade-off is controlling substitutions and maintaining uniform behavior across multi-source components, which should be governed by explicit rules and verified by repeatable acceptance testing.
Application-Specialized Categories
Surgical and endoscopy-focused manufacturers are best when your product must behave predictably through complex signal chains and operational triggers such as switching, movement, and integration with intermediate devices. Validation should focus on stability under realistic chain conditions rather than isolated bench tests.
Diagnostic-focused manufacturers are best when your program depends on calibration discipline, unit-to-unit uniformity, and QC repeatability. The priority is not only initial performance but also long-term drift behavior and support for re-validation workflows over the product lifecycle.
OEM due diligence questions that reveal real capability
Strategic due diligence questions expose actual manufacturer capabilities beyond marketing materials and surface-level presentations.
The fastest way to separate mature OEM partners from “spec-sheet suppliers” is to ask questions that force evidence: traceability proof, change control rules, repeatable acceptance, and lifecycle commitments.
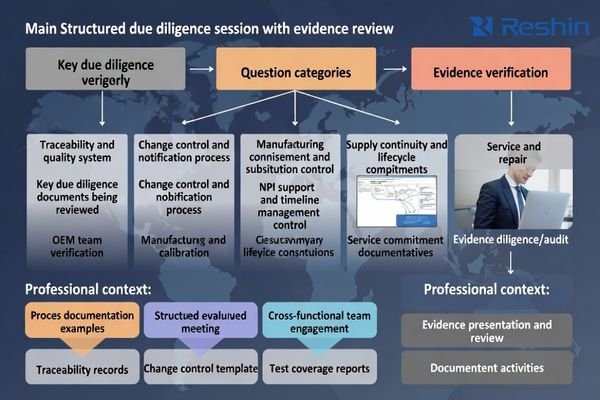
Use these questions as a practical RFI/RFQ and pilot checklist:
- How do you trace a field issue to a batch and document corrective action?
- What is your PCN/ECN process6, typical lead time, and approval workflow?
- How do you control component substitutions and validate impact?
- What does your factory calibration and outgoing test coverage look like?
- How do you demonstrate unit-to-unit consistency across different production periods?
- What NPI support do you provide for EVT/DVT/PVT, and who owns timeline escalation?
- What are your commitments for key components and long-term supply continuity?
- What is your spare parts policy and typical repair turnaround?
- How do you handle end-of-life notices and migration paths for long-lived programs?
- What documentation artifacts will you provide for acceptance and future re-validation?
A strong partner answers with process clarity and evidence examples, not vague assurances. The goal is to confirm you can operationalize the relationship through repeatable testing, predictable change communication, and service policies that match your deployment reality.
Pilot strategy: how to validate manufacturers in real environments
Comprehensive pilot programs provide reliable validation of manufacturer capabilities under actual deployment conditions.
An effective pilot validates stability, consistency, documentation usability, and support responsiveness in the same mounting, cabling, and operational conditions your product will face after launch.
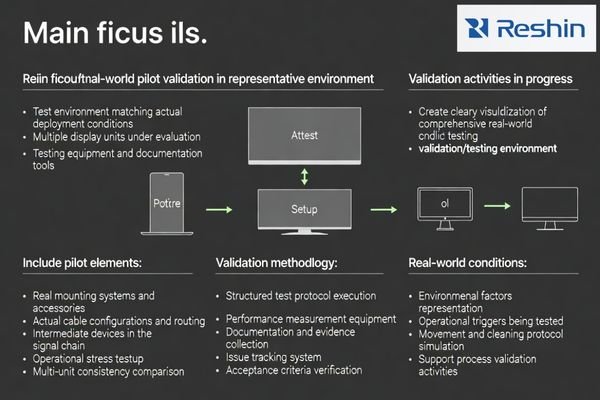
To keep pilots actionable, limit scope but increase realism. Use identical mounting systems, cable configurations, and intermediate devices, and include operational triggers7 such as switching, movement, and routine cleaning. Validate multiple units, not just one, because OEM risk often shows up as variability rather than absolute failure.
Pilot output should include repeatable acceptance results, a documented issue log with closure behavior, and a version baseline that defines what was validated. Just as importantly, measure support: response time, escalation clarity, and the manufacturer’s ability to explain root cause and corrective action when problems appear.
Red flags and common OEM pitfalls
Recognizing warning signs prevents costly partnerships with manufacturers who cannot deliver reliable OEM support throughout project lifecycles.
Red flags include prototype-to-production drift without disciplined change control, certificates without usable documentation, weak traceability evidence, unclear supply continuity, and service gaps that increase downtime and total cost.
Watch for patterns that predict long-term pain: sample units that look good but do not match production builds, substitutions that occur without transparent notice, and documentation that cannot be used for acceptance or audits. Also treat vague EOL policies as a risk, because OEM programs often outlive component availability without warning unless lifecycle governance is explicit.
Conclusion
Top medical display manufacturers for OEM projects should be defined by best-fit evidence rather than universal rankings. A systematic approach—shortlisting by OEM-fit category, demanding traceability and change control evidence, and validating through a realistic pilot—reduces risk across development and market deployment.
Our experience at Reshin shows that OEM success improves when evaluation focuses on operational controls, documentation discipline, and predictable support, not just initial samples. With clear gates, repeatable acceptance, and a change-control agreement, OEM teams can scale confidently while keeping field behavior stable over the product lifecycle.
✉️ info@reshinmonitors.com
🌐 https://reshinmonitors.com/
-
Exploring systematic evaluation methods can enhance your risk management strategies in product development and deployment. ↩
-
Understanding OEM risks is crucial for ensuring product reliability and performance in the field. ↩
-
Exploring strategies for supply continuity can help manufacturers mitigate risks and enhance their supply chain resilience. ↩
-
Understanding decision artifacts is crucial for managing risk in OEM programs effectively. ↩
-
Explore this link to understand how Customization-Focused manufacturing can enhance your product development process. ↩
-
Understanding the PCN/ECN process is crucial for managing changes effectively and ensuring product quality. ↩
-
Understanding operational triggers can enhance pilot training and improve safety protocols in aviation. ↩