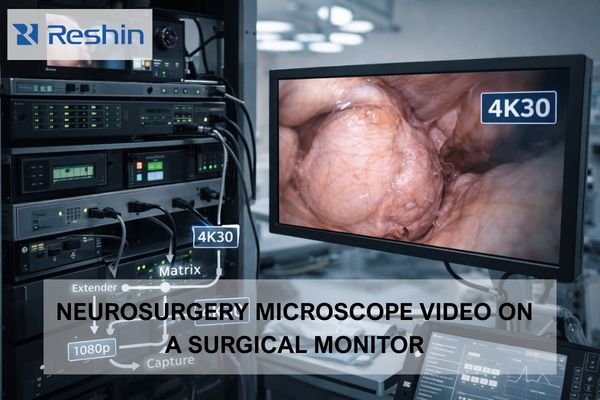
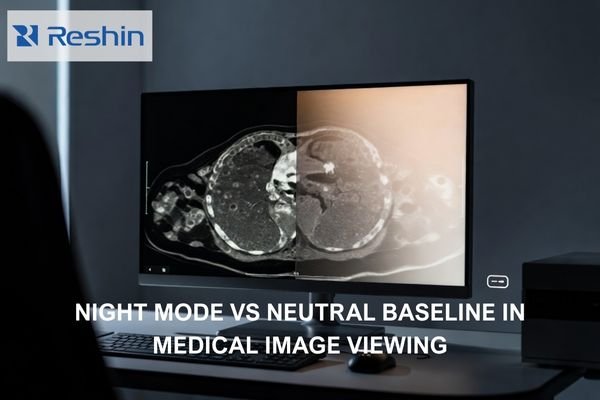
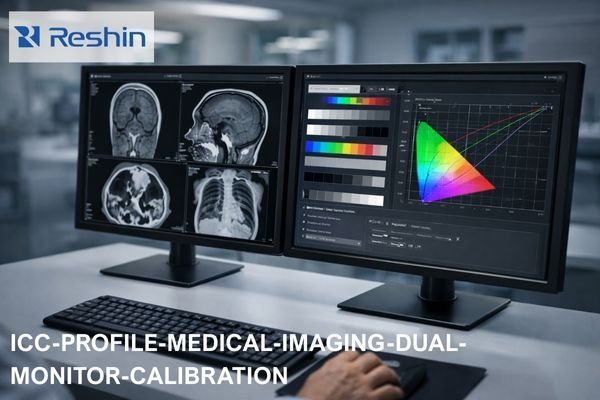
What Core Documents Are Needed for CE Marking of medical grade monitors?
Core CE documents typically include: device description and variants, a risk management file, a requirements-to-evidence matrix, safety/EMC and performance verification reports, controlled labeling/IFU, and PMS/PMCF/vigilance plans. Delays most often come from inconsistent claims across marketing, labeling, and evidence, or missing traceability from requirements to test reports.