In medical display projects, the phrase “registration support” often gets used too loosely. Many buyers hear it and assume the supplier will somehow take over the full regulatory path. In actual projects, that is usually not how it works. From the supplier side, the more useful role is to help make the preparation process clearer, better structured, and easier to keep under control.
Manufacturer-side registration support in medical display projects is not about the supplier completing registration on the buyer’s behalf. It is a structured form of compliance and registration support built around requirements alignment, controlled documentation, verification cooperation, technical clarification, and disciplined change management, so the buyer’s own registration preparation can move forward with fewer blind spots and less avoidable rework.
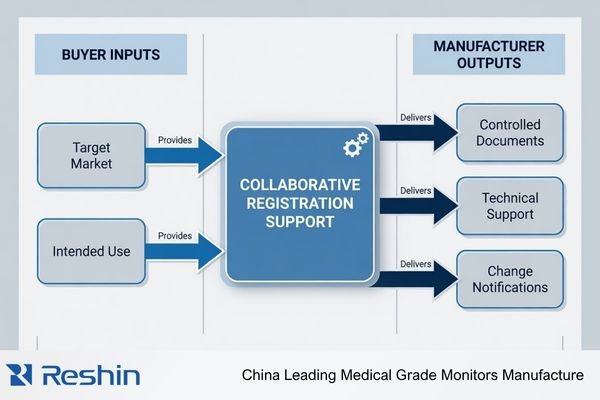
When I begin this kind of project with a partner, I usually start by defining the boundary before anything else. That sounds basic, but it saves time later. The manufacturer side is there to provide product-specific documentation, technical clarification, verification cooperation, and controlled updates in a way the project team can actually use. The manufacturer side is usually not the legal owner of the registration itself.
That distinction matters because good support is not measured by how many files are sent in the first week.1 It is measured by whether the information stays relevant to the actual configuration under review, whether technical questions can be answered without ambiguity, and whether later changes are handled in a controlled way. In practice, strong support makes the buyer’s work more manageable. It does not replace the buyer’s responsibility.
Manufacturer-Side Registration Support Is Not the Same as “The Supplier Handles Registration”
In medical display programs, one of the most common misunderstandings I see at the beginning is the idea that “registration support” means the supplier will run the submission process. That assumption usually creates confusion later unless the roles are separated early.
In real-world programs, the manufacturer side usually supports the project through product documentation, technical clarification, verification cooperation, and change communication. Registration ownership, market-entry responsibility, and formal submissions typically remain with the buyer’s local legal entity or designated regulatory role.

From a project point of view, that boundary is useful, not restrictive. The supplier knows the product, the configuration logic, the test background, and the revision history. The buyer or its regulatory team knows the target market, the submission route, and the local approval obligations. When both sides stay in the part they can actually control, cooperation becomes more efficient2.
In our own projects, I usually describe the supplier role in practical terms: we help make the product side of the story clearer. That may include preparing controlled technical files, explaining how a feature or claim should be interpreted, supporting sample-related verification questions, and communicating later changes that could affect the project. The buyer side then decides how that information is used in formal preparation and submission.
Registration Support Usually Starts with Requirements Alignment, Not with Document Delivery
From an engineering standpoint, I do not treat registration support as a simple document request. The first useful step is usually to align on what the project is actually trying to achieve.
Effective registration support usually starts with requirement alignment: target market, intended use boundary, current project stage, and the real purpose of the request, whether that is internal review, registration preparation, or third-party verification.

A lot of avoidable delay comes from requests that are too broad at the beginning3. When a buyer asks for “all compliance documents,” the request sounds comprehensive, but it often produces a package that is only partly useful. In practice, I usually need a much narrower picture first. Which market is involved? What role will the display play in the final system? Are we supporting an early internal review, a formal registration-preparation stage, or a test-related clarification exercise?
The Inputs We Normally Confirm First
In our own project reviews, I usually confirm four things before we start assembling a support package: the target market, the intended-use boundary, the current project stage, and the exact reason the buyer needs support at that moment. Those inputs sound simple, but they decide whether the next step should be configuration definition, documentation packaging, sample coordination, or clarification of test assumptions.
Why Early Alignment Saves Rework
When those conditions are clear early, the support becomes much more focused. The document set is easier to prepare, the explanations are easier to keep relevant, and the buyer’s team spends less time sorting through material that does not match the real question in front of them. In my experience, that early alignment often does more for project speed than rushing to send a large file package too soon.
Documentation Support Works Best When It Is Controlled, Structured, and Product-Specific
In real projects, the value of documentation support is not decided by how many files the supplier can send. I pay much more attention to whether the documents are controlled, revision-aware, and tied to the actual product configuration being reviewed.
Documentation support becomes genuinely useful when it is accurate, clearly versioned, and aligned with the evaluated sample or intended production configuration. If the document set drifts away from the real project baseline, the buyer is left carrying extra work and unnecessary risk.

The key issue is alignment. If the paperwork describes one condition, the sample reflects another, and the later supply follows a third, the buyer’s team has to spend time reconciling differences that should not have existed in the first place4. That is where support stops being helpful and starts becoming another layer of project management burden.
The table below shows the difference between loosely structured documentation support and support that is more usable in a registration-related project.
| Aspect of Support | Less Controlled Support | Better-Controlled Support |
|---|---|---|
| Accuracy | Generic files are provided that only partly reflect the specific model or configuration under review. | Documents are tied to the actual model and revision being used for the project. |
| Revision Control | Files are shared without clear version logic, making later updates harder to track. | Document revisions are visible and easier to follow across the project timeline. |
| Synchronization | The sample, the documents, and later supply do not stay fully aligned. | The document set stays closely matched to the evaluated configuration and later project communication. |
On our side, I usually try to keep the document package tied to the same product baseline the project is actually discussing. That sounds obvious, but it is one of the first things that breaks down when support is treated as a one-time file handover instead of an ongoing controlled process.
Sample, Verification, and Technical Clarification Are a Core Part of Registration Support
Many buyers start by thinking registration support is mostly paperwork. In actual programs, the more important part is often the work around the paperwork: sample confirmation, verification discussion, and technical clarification.
Before registration preparation can move forward with confidence, the project usually needs a clear baseline configuration, a workable interpretation of key product claims, and alignment on test assumptions or evaluation boundaries. That part of the support is often more useful than a document package by itself.

A document package only becomes meaningful when both sides are talking about the same product condition. That is why sample-related discussion matters. The project often needs to confirm which configuration is being evaluated, whether the stated performance is being understood under the right assumptions, and whether the current documentation really matches the sample in front of the buyer’s team.
Establishing the Baseline
The first practical step is usually to confirm the baseline that later discussion will refer to. In our own work, I normally want that baseline to be explicit enough that later questions can point back to one clear reference. That includes the model, the current revision status, any relevant firmware condition, and the accessory or configuration scope that matters to the evaluation.
Clarifying Claims and Verification Boundaries
The next step is usually to remove ambiguity around claims and test conditions. If the project is using certain technical statements, both sides should understand what those statements actually mean in context. I have found that many later misunderstandings can be avoided when the team writes down a few simple points early: which sample is the reference, which claims are being relied on, which assumptions sit behind the verification, and what still needs clarification before the project moves deeper into preparation.
Controlled Change Management Is What Keeps Registration Preparation Stable Over Time
In regulated medical display projects, I place almost as much weight on controlled change management as on the initial support package. Early cooperation may look strong, but if later revisions are not managed properly, the usefulness of the earlier work drops quickly.
If manufacturer-side support stops after the initial document handover and does not continue through revision awareness, change communication, and controlled updates, the support is incomplete. What matters most is support that remains usable before, during, and after change.
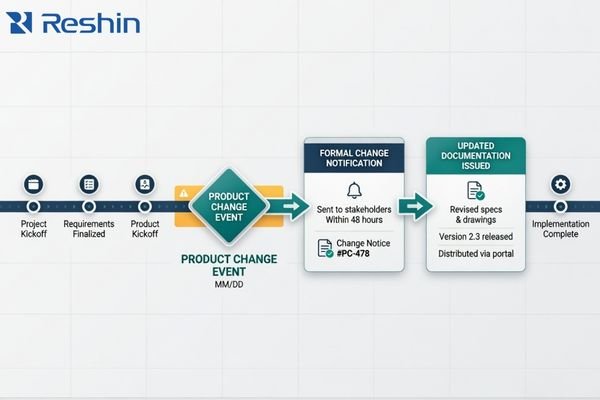
Registration-related preparation often takes time. During that time, a sample version may change, a document may be updated, or a product detail may need clarification. When those changes are visible and managed, the project can usually absorb them. When they are not, earlier preparation starts to lose continuity.
This is why I do not view the first document package as the end of support. It is only the first stage. The more important question is whether the supplier can continue to communicate revision status in a disciplined way after the initial handover. In our own projects, I usually treat ongoing revision awareness as part of the support itself, not as an optional extra, because that is what helps keep earlier verification and documentation work usable later on.
FAQ About Manufacturer-Side Registration Support in Medical Display Projects
Does manufacturer-side registration support mean the supplier will complete registration for the buyer?
Usually not. In most projects, the supplier supports the product side of the preparation through documentation, technical clarification, verification cooperation, and controlled change communication. Registration ownership and market-entry responsibility still remain with the buyer’s legal entity or designated regulatory role.
What should buyers confirm first before requesting compliance or registration support?
I usually recommend confirming the target market, the intended-use boundary, the current project stage, and whether the request is meant for internal review, registration preparation, or third-party verification. Once those points are clear, the support becomes much more relevant.
Is registration support mainly a document task?
Not entirely. Documentation matters, but it works much better when it is supported by requirement alignment, technical clarification, sample coordination, and controlled change communication. Without those pieces, the file package alone often does less than people expect.
Why does change management matter so much in registration-related projects?
Because once the sample, configuration, or document revision changes without being communicated clearly, earlier preparation work can lose continuity. That usually means more rechecking, more internal coordination, and more pressure on the timeline.
Good Registration Support Does Not Remove Responsibility — It Improves Project Control
The real value of manufacturer-side registration support is not that the supplier takes over responsibilities that belong somewhere else. It is that the supplier handles its own side of the cooperation in a way that is clearer, more controlled, and more usable for the buyer’s team.
In medical display projects, that usually means structured requirement alignment, controlled documentation, sample and verification cooperation, technical clarification, and disciplined change management. If your project is moving toward registration preparation, the more useful next step is usually not to ask whether the supplier will “handle registration,” but whether the supplier can provide the kind of Compliance Support that keeps document alignment, clarification, and change control manageable as the project moves forward.
✉️ info@reshinmonitors.com
🌐 Compliance Support
-
"Quality in project management–a practical look at chapter 8 of … – PMI", https://www.pmi.org/learning/library/practice-three-project-quality-management-7198. Project Management Institute (PMI) standards recommend assessing vendor or supplier support based on the clarity, relevance, and adaptability of information over time, rather than initial documentation volume. Evidence role: general_support; source type: institution. Supports: Good support is not measured by how many files are sent in the first week.. Scope note: These are general project management guidelines and may require tailoring for specialized technical or regulatory projects. ↩
-
"Stakeholder diversity and collaborative innovation: Integrating the …", https://www.sciencedirect.com/science/article/pii/S0148296323003132. Empirical studies in project management report that clear delineation of stakeholder roles and responsibilities is linked to higher collaboration efficiency. Evidence role: general_support; source type: paper. Supports: When both sides stay in the part they can actually control, cooperation becomes more efficient.. Scope note: Findings are primarily based on software and engineering projects and may vary in other sectors. ↩
-
"Caution! Bad Project Requirements Will Cause Delays – Reqtest", https://reqtest.com/en/knowledgebase/bad-project-requirements-will-cause-delays/. Empirical studies indicate that projects with unclear or overly broad initial requirements experience significantly higher delay rates, with average schedule overruns of up to 30%. Evidence role: statistic; source type: paper. Supports: A lot of avoidable delay comes from requests that are too broad at the beginning.. Scope note: Most data derive from software development studies and may not fully generalize to all regulatory compliance or hardware integration projects. ↩
-
"Right-size your quality management documentation for SQMS No. 1", https://www.journalofaccountancy.com/issues/2025/oct/right-size-your-quality-management-documentation-for-sqms-no-1/. Quality and configuration management studies report that discrepancies among documentation, prototypes, and production units can increase reconciliation tasks by up to 15–30%, raising overall project management effort. Evidence role: statistic; source type: paper. Supports: Misalignment between paperwork, samples, and supply increases the buyer’s team workload due to reconciliation of discrepancies.. Scope note: The data are drawn from manufacturing case studies and may not precisely reflect registration-related projects. ↩