Evaluating a medical display manufacturer’s certifications requires focusing on evidence that directly supports medical quality control, product compliance, and documentation readiness.
When evaluating a medical display manufacturer from a distributor’s perspective, the focus should not be on the quantity of certificates. The more important task is to identify which certifications and compliance documents actually support medical quality control, product compliance, and documentation readiness. In most cases, ISO 13485, product-related compliance information, and evidence of document traceability matter far more than general company awards or patent counts.

In my work supporting distributor qualification and supplier review, I often find that a manufacturer’s certification profile reveals much more than its marketing materials do. A long list of general awards may create a positive first impression, but it does not provide the same level of assurance as a current ISO 13485 certificate1 supported by clear, product-related compliance information. For distributors and local partners, the real value lies in certifications and documents that indicate a manufacturer is prepared for documentation review, audits, and long-term cooperation in medical markets. Focusing on the right evidence early is a practical risk-control step because it helps distinguish between manufacturers that are prepared for regulated business requirements and those that mainly present surface-level credentials.
Why Certification Review Matters Before Distributor Cooperation Starts
Certification review is not an administrative checkbox; it is a foundational step in supplier due diligence and risk management for distributors.
From a distributor’s perspective, certification review is an early filter used to identify manufacturers that may create downstream problems in documentation support, customer audits, public tenders, market-entry preparation, and long-term supply consistency. It is not the full supplier evaluation, but it is often the first gate in deciding whether a manufacturer is worth deeper commercial and operational review.

Based on the distributor qualification work I support, the main purpose of reviewing a manufacturer’s certifications is to assess whether that supplier is aligned with the practical demands of medical business, not just whether it can produce a working sample. A manufacturer without the right quality-system foundation2 and product-related compliance evidence may still be able to deliver a functional monitor, but it may struggle when documentation must be updated, when a customer requests supporting files, or when a project requires more formal review. For a distributor, that gap can create commercial risk, project delays, and unnecessary friction. That is why I treat the initial certification check as an indicator of whether a manufacturer is operating within a medical-quality framework that can support serious long-term cooperation.
Which Certifications Should Be Treated As Decision-Critical
From an engineering and risk management standpoint, not all certifications carry equal weight in predicting a manufacturer’s suitability for medical projects.
Decision-critical certifications for a medical display manufacturer can usually be grouped into three categories: first, ISO 13485 for medical-device-oriented quality management; second, publicly stated CE / MDR-related compliance information relevant to the product line and target market; and third, publicly stated compliance with standards such as IEC 60601-1 for product safety in medical environments. These are more relevant to initial screening because they are directly connected to quality control, product suitability, and documentation readiness.

When I evaluate a potential manufacturing partner, I prioritize certifications and compliance information that are directly tied to the product lifecycle and to the distributor’s documentation responsibilities. General company honors may still be useful as background signals, but they should not lead the decision. The more important question is whether the manufacturer can show evidence that its products are built within a controlled quality system and supported by product-related compliance documentation that can be reviewed and maintained over time.
ISO 13485: Medical Device Quality Management System
This is often the most important quality-system credential in an initial medical manufacturer review. Unlike broader quality standards, ISO 13485 is specifically associated with medical-device-oriented quality management. It is relevant because it points to more disciplined control over design, production, records, traceability, and change handling. A manufacturer with a mature ISO 13485 system is generally better positioned to respond to documentation requests, manage controlled product changes, and support the level of consistency expected in medical business.
CE / MDR and Other Market-Relevant Compliance Documentation
For distributors working across regulated markets, publicly stated [CE / MDR](https://decomplix.com/medical-device-ce-marking-guide/)3-related compliance information is highly relevant, but the visible mark itself should never be the only checkpoint. The more important issue is whether the manufacturer can connect its public compliance claims to the specific product family, provide model-relevant supporting information, and maintain documentation when customers ask questions. In practical terms, what matters is not simply whether a mark appears on a product or brochure, but whether the manufacturer can support that claim with organized, product-related documentation.
IEC 60601-1: Medical Electrical Equipment Safety
For medical displays used in clinical environments, publicly stated compliance with standards such as IEC 60601-1 is an important signal because it relates to electrical safety and use in medical settings. From a distributor-evaluation standpoint, this matters less as a theoretical standard and more as a practical screening question: can the manufacturer show that safety-related compliance is part of its documented product framework rather than only a marketing statement? That distinction is important because distributors often need confidence that the product line has been developed and reviewed with medical-use conditions in mind.
Why ISO 13485 Usually Matters More Than General Management Certificates
In supplier evaluations, it’s common to see a mix of ISO certificates, but not all of them signify the same level of readiness for medical device manufacturing.
While a standard like ISO 9001 indicates a general quality management framework, ISO 13485 is usually more important for medical projects. That is because ISO 13485 is more directly aligned with the documentation discipline, traceability expectations, change control, and product-related quality controls that distributors typically rely on when working with medical products over the long term.

From a supplier-review standpoint, I often find that buyers who are new to medical sourcing can place different ISO certificates at roughly the same level. In practice, the distinction is significant. An ISO 9001-certified factory may still be well managed, but that alone does not indicate the same level of medical-product discipline that distributors usually need to see. For example, when a project depends on stable product configuration, clearer traceability, and more structured change communication, ISO 13485 is generally the stronger signal.
| Aspect | ISO 9001 (General Quality Management) | ISO 13485 (Medical Device Quality Management) | Why It Matters for Distributor Qualification |
|---|---|---|---|
| Focus | General quality management across many industries. | Quality management aligned more closely with medical-device-related expectations. | Helps indicate whether the manufacturer’s system is structured for medical-market cooperation rather than only general industrial production. |
| Risk Management | Broad risk-based management thinking. | More formal attention to risk, control, and product-related quality discipline. | More relevant when distributors need confidence in product stability and controlled processes. |
| Documentation | Controlled documentation is required. | Stronger emphasis on documentation discipline and traceability in medical-product contexts. | Important for audits, tenders, customer reviews, and requests for supporting files. |
| Change Control | Changes are managed, but usually in a more general framework. | Change control is treated with greater discipline in medical-oriented quality systems. | Helps reduce the risk of undocumented product changes that can affect supply, documentation, or customer acceptance. |
When I support a distributor in screening a new manufacturing partner, the presence of ISO 134854 is usually a stronger indicator of medical-project suitability than a collection of more general management certificates. It does not answer every question by itself, but it does suggest that the manufacturer is operating with a level of process discipline that is more compatible with regulated medical business.
Why A CE Mark Alone Is Not Enough For Distributor Evaluation
Seeing a CE mark on a product is a common starting point for evaluation, but for a distributor, it should never be the conclusion.
Relying only on a visible CE mark is a common mistake. A more reliable review checks whether the underlying compliance information aligns with the specific product models being considered, whether the documentation appears current and maintainable, and whether the manufacturer can provide clear support when compliance-related questions arise. The issue is not simply whether a CE mark exists, but whether the compliance claim behind it is organized, product-relevant, and supportable.

Based on the distributor qualification work I have seen, the more important issue is always the substance behind the mark. A practical risk appears when a distributor assumes that one public symbol or one broad statement automatically covers every model under discussion. Another common problem is that public compliance claims are available, but the supporting information is not well organized when the project moves into deeper review. In that kind of situation, the logo itself does not solve the distributor’s problem. What matters is whether the manufacturer can connect the visible claim to product scope, documentation readiness, and continued support.
Why Patents And Awards Should Not Replace Quality-System Evidence
Patents, industry awards, and other company honors can be positive signals, but they should be viewed as supplementary information, not as primary evidence of manufacturing capability.
From a risk-assessment perspective, patents and awards may indicate R&D effort, industry participation, or a degree of recognition. However, they do not prove that a manufacturer has a stable, controlled, and medical-project-ready production system. These credentials can support confidence, but they should not replace stronger evidence such as ISO 13485, product-related compliance information, and document traceability.

When I review a potential supplier, I treat patents and awards as context rather than as decision proof. They may help show that the company has invested in development, maintains technical differentiation, or has received industry recognition. That information is useful, but it does not answer the distributor’s most important questions. It does not confirm whether the manufacturer can maintain consistent product quality, respond quickly with documentation, or manage controlled product changes in a disciplined way. In supplier evaluation, those practical capabilities usually matter more than recognition signals alone.
What Good Certification Support Looks Like In Practice
A reliable manufacturer should not only display certificates but also show that its certification-related support works in real distributor communication.
In practice, good certification support means the manufacturer can explain what a certificate covers, connect public compliance claims to the relevant product line, provide supporting information in an organized way, and respond with reasonable clarity when distributors request documentation. A certificate becomes much more meaningful when it is backed by usable documentation, clear scope explanation, and disciplined follow-up.

From a distributor-evaluation perspective, this is often the point where stronger and weaker manufacturers begin to separate. A weaker supplier may show a certificate quickly but struggle to explain which products it covers, how public compliance statements relate to current models, or how documentation is maintained when customer questions arise. A more prepared manufacturer usually does several things better. It can explain certification scope5 in a way that matches the product category under discussion. It can provide public supporting information without creating confusion between product families. It can respond in a structured manner when a distributor requests clarification. Just as importantly, it treats documentation support as part of the business relationship rather than as a one-time administrative burden.
For distributors and local partners, this practical layer matters because certificates alone do not move a project forward. What supports tenders, customer review, and long-term cooperation is the manufacturer’s ability to connect those certificates to real products, real files, and real communication. That is why I do not treat certification review as a paper exercise. I treat it as an early test of whether a manufacturer is likely to be workable in actual medical business.
What Distributors Should Verify Beyond The Certificate Itself
A certificate is a static document. The real test of a manufacturer’s capability lies in the dynamic processes and support systems that surround it.
For distributors, a certificate is only an entry condition. The more important step is to verify whether the certification scope matches the product category, whether the document appears current, and whether the manufacturer can provide timely documentation support when questions arise. A more reliable partner usually demonstrates consistency not only through certificates, but also through responsiveness, document control, and stable cooperation over time.
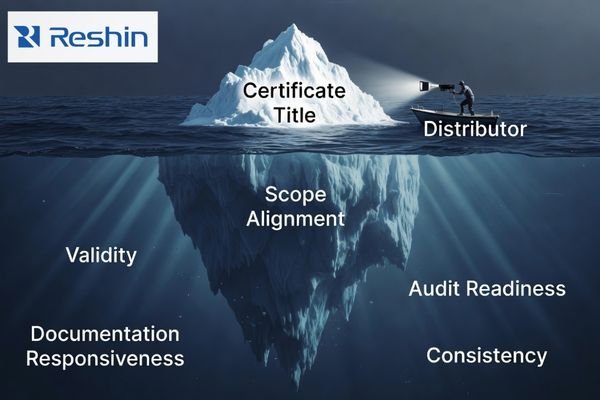
In supplier qualification, the difference between a weaker and a stronger partner often becomes clear after the certificate is shown. A stronger partner can explain how the public certification relates to the product line, provide supporting files in an organized way, and respond with reasonable speed when a distributor needs product-specific clarification. Here are the points I usually recommend verifying:
- Scope Alignment: Does the certificate or public compliance statement clearly relate to the product category you plan to distribute?
- Validity: Does the documentation appear current, and can the manufacturer explain its update status when asked?
- Documentation Responsiveness: Can the manufacturer provide relevant supporting information clearly and within a reasonable timeframe?
- Audit and Tender Readiness: Is there evidence that the manufacturer can support document requests tied to customer review, qualification, or tender preparation?
- Consistency Across Models: Do the product lines under discussion appear to be managed under a consistent quality and compliance framework?
Verifying these practical points usually gives a distributor a clearer picture of real partnership readiness than reading the certificate title alone.
How To Use Certifications As Part Of A Broader Supplier Evaluation
In real-world partnership decisions, certifications should not be used as a standalone decision tool but as a foundational component of a comprehensive evaluation framework.
The more reliable approach is to place certification review inside a broader supplier-evaluation process. In practice, that means first confirming the decision-critical certifications, then assessing how well the manufacturer supports documentation requests, and then reviewing factors such as product-line clarity, change-control discipline, communication responsiveness, and long-term cooperation stability. From my perspective, the most dependable manufacturer is rarely the one with the longest list of certificates. It is more often the one that combines a credible quality framework, organized product-related documentation, practical responsiveness, and consistent cooperation capability into one controlled operating model.
From a distributor perspective, certifications should therefore be treated as the starting point of supplier evaluation, not the final answer. They are useful because they help narrow the field and reveal whether a manufacturer is likely to support regulated medical business in a disciplined way. But the final decision should still depend on how well that manufacturer performs when documentation is requested, products are reviewed, and cooperation moves from initial contact into real project execution.
✉️ info@reshinmonitors.com
🌐 https://reshinmonitors.com/
-
Understanding ISO 13485 certification is crucial for ensuring compliance in medical markets, making this resource invaluable for informed decision-making. ↩
-
Exploring this topic will provide insights into the essential systems that ensure product safety and compliance. ↩
-
Exploring CE and MDR compliance helps manufacturers navigate regulations and ensure their products meet market standards. ↩
-
ISO 13485 is essential for ensuring quality in medical devices, making it vital for distributors to understand its importance. ↩
-
Understanding certification scope is crucial for evaluating manufacturers and ensuring compliance with industry standards. ↩