For many first-time buyers, sourcing a medical display from China begins with two very practical questions: What is the price, and how fast can it ship? Those questions matter, but in my experience they rarely decide whether the cooperation turns out well. What usually matters more is whether the supplier can stay consistent from sample review to documentation, production, and later replenishment.
When buying a medical display from China for the first time, the safest approach is to confirm six things early: the exact application scope, alignment between sample and production, documentation support, OEM/customization boundaries, supply continuity, and communication quality. A capable medical display manufacturer should be able to support all six, not just provide a competitive first quotation.

In the projects I support with PACS and KVM partners, I have seen first-time sourcing move smoothly when these basics were clarified early, and I have also seen it become much harder than it needed to be when they were not. Buyers naturally look at MOQ, lead time, and pricing1 first. The better question is whether the supplier can stay predictable as the project moves from inquiry to sample, from sample to production, and from the first shipment to repeat orders.
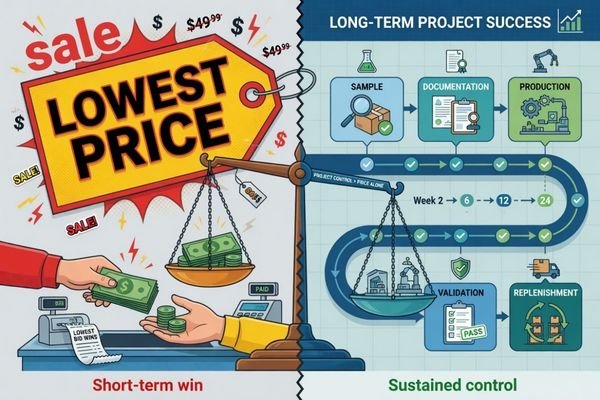
A low opening quote can still turn into an expensive project if the sample does not match later production, if the document package is incomplete, or if replenishment becomes unstable after the first order. From an engineering standpoint, the real value is not simply finding the lowest-cost vendor. It is finding a supplier that stays controllable, responsive, and steady through the full cooperation cycle.
Buying a Medical Display from China for the First Time Is Not Mainly a Price Decision
When buyers work with a Chinese medical display supplier for the first time, price is usually the easiest thing to compare. It is visible, immediate, and simple to put into a spreadsheet. The harder part is judging what happens after the quotation is accepted.
For a first-time buyer, the more important evaluation is whether the supplier can keep samples, documentation, production, and communication under control. A low initial price can lose its value quickly if the project later faces specification drift, weak support, inconsistent replenishment, or uncontrolled revisions.
In my experience, the projects that get difficult are usually not the ones that started with a slightly higher unit price. They are the ones where the approved sample and the delivered product stop matching, where documents become difficult to track, or where routine questions take too long to close. That is where extra engineering time, delayed approvals, and internal friction start to appear.
That is why first-time sourcing2 is better treated as risk control, not just purchasing. Price matters, but the first shipment is only one point in a much longer project timeline. In our own work, we usually tell new partners that the first quotation is only one part of the conversation. Before we treat a project as ready, we want both sides to be clear on product scope, document expectations, approval flow, and future supply logic.
Confirm the Exact Application Scope Before Requesting a Quote
The phrase “medical display” sounds specific, but in real projects it covers very different use cases. A display for radiology review, a display for surgery, and a display used inside an equipment interface may all sit in the same broad category while requiring very different performance and support.
Before requesting a quote, buyers should define the application clearly: system type, use environment, signal input, target size, resolution, mounting method, expected quantity, and acceptance criteria. Without that information, even a detailed quotation can still point to the wrong product direction.
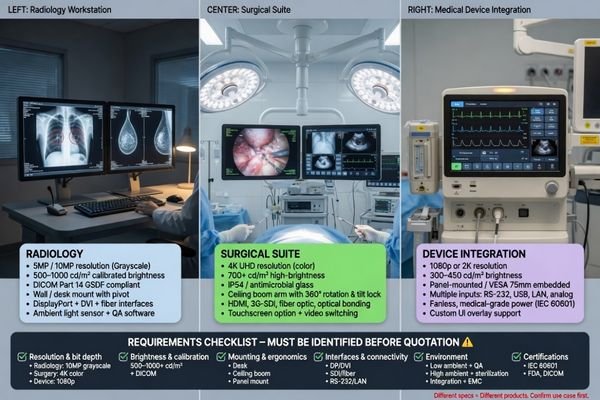
When I receive a first inquiry, the first thing I need to understand is the role of the display inside the system. A request for a “24-inch medical monitor3” is not enough on its own. I need to know whether the project is diagnostic, surgical, clinical review, ultrasound, equipment integration, or something more customized. I also need to know what signal comes in, where the display will be installed, and what the buyer expects it to do in daily use.
That is why I usually encourage buyers to send the application boundary first, not just the target size. A more complete starting brief leads to a much more useful response, whether the next step is a standard product recommendation or an OEM medical display project.
In our own project reviews, we usually begin with a small group of practical inputs: application type, signal source, size expectation, mounting condition, and target quantity. That sounds basic, but it often decides whether a project starts on the right path or spends two weeks discussing the wrong product family.
Confirm That Sample, Specification, and Mass Production Will Stay Aligned
One of the biggest concerns for a first-time buyer is whether the approved sample will really represent the product that is later delivered at scale. That concern is completely reasonable. A sample that looks good has limited value if later production drifts away from the validated version.
Buyers should confirm that the sample, the specification sheet, and later production are aligned under clear revision control. The key question is not just whether a sample can be sent, but whether that sample reflects a stable production plan and a documented change-notification process.
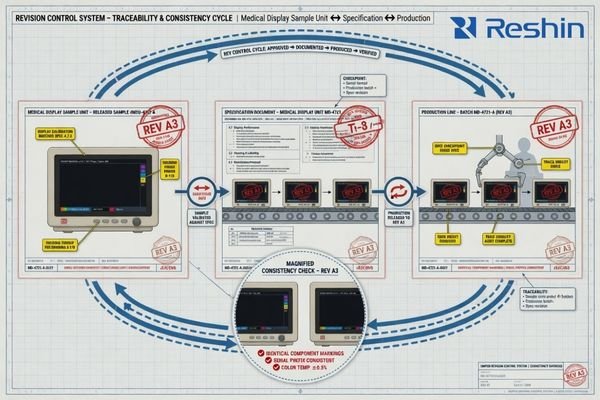
When I review projects that ran into trouble, the problem often appears after sample approval4 rather than before it. A later delivery may use a different panel source, a changed interface board, another default setting, or a revised document that was never clearly communicated. Each change may look minor on its own, but together they can disrupt validation, installation, or customer-facing delivery.
For a first-time buyer, a few simple confirmations go a long way: Is the sample built from components intended for stable production planning? Is the specification sheet under revision control? How are later changes communicated? What happens if a key component reaches end-of-life? These questions help distinguish a short-term sample supplier from one that can actually support a real program.
In our team, this is one of the first areas we try to make clear to new partners. When a sample is under review, we usually clarify what version it represents, which document it is tied to, and how later changes would be communicated if a component or board needs adjustment. That kind of early alignment often prevents much bigger problems later.
Confirm Documentation and Certification Support Beyond the Certificate Name
It is easy to ask a supplier whether they have ISO or CE. It is usually more useful to ask what documentation they can actually provide for the project in front of you.
For first-time buyers, the real confirmation point is not the certificate name alone, but whether the supplier can provide a project-relevant document package, keep it consistent through revisions, and support internal review, local registration preparation, or customer approval when needed.

From an engineering standpoint, a certificate is only the beginning of the conversation. What a project usually needs is a usable document package: specification files, declaration-related documents, applicable standards information, version records, and other supporting notes that help the buyer’s engineering, quality, or compliance team move the project forward.
A supplier’s ability to keep those materials relevant, traceable, and current is often a better sign of project readiness than the certificate itself. For a first-time cooperation, strong documentation support5 shows that the supplier is prepared to work within a structured project environment, not simply ship a product.
In our own projects, new buyers often begin by asking for one certificate, but the real discussion quickly moves to the surrounding files and how they match the exact product under review. That is usually where trust starts to build. A supplier that can explain what is available, what is product-specific, and how version control is handled is much easier to work with over the long run.
Confirm What “OEM” or “Customization” Really Means Before the Project Moves Forward
The words “OEM” and “customization” sound straightforward, but in practice they often mean very different things depending on the supplier. For first-time buyers, that gap in interpretation can create unnecessary confusion later.
Before the project moves forward, buyers should confirm whether customization means branding only, or whether it also includes interfaces, housing, stands, labels, packaging, default settings, and deeper engineering adjustments. Just as important, they should confirm how those changes affect MOQ, sample timing, validation flow, lead time, and future replenishment.
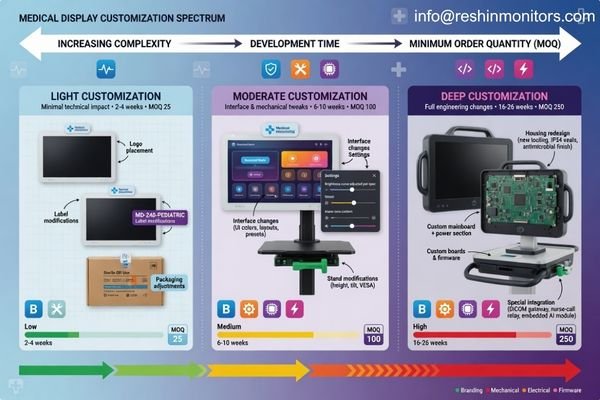
When I discuss customization with a new partner, I try to define the scope in very practical terms. A bezel logo change, a custom label, a special carton, a stand adjustment, and a redesigned enclosure may all fall under “OEM” in casual conversation, but they do not carry the same project impact. Some are simple branding tasks. Others become engineering tasks with real implications for schedule, testing, and long-term version control.
That is why first-time buyers should not stop at asking whether OEM is supported. The better question is how far the customization can go while still remaining stable in documentation, production, and repeat orders.
In our own workflow, we usually separate lighter customization from deeper engineering changes very early. That makes it easier for both sides to understand what can move quickly, what needs formal evaluation, and what will affect timing, validation, or future continuity. For a first-time buyer, that clarity can prevent a lot of wrong assumptions.
Confirm Supply Logic, Lead Time, and Reorder Continuity Before You Approve the First Order
The first shipment matters, but it is rarely the end of the story. In most serious projects, the real test of a supplier starts after the first order has already been approved.
Before confirming the first order, buyers should establish the broader supply logic: sample lead time, pilot timing, mass-production cadence, MOQ for repeat orders, replenishment expectations, and the supplier’s approach to version continuity. Predictable replenishment often matters more than the price of the first shipment.
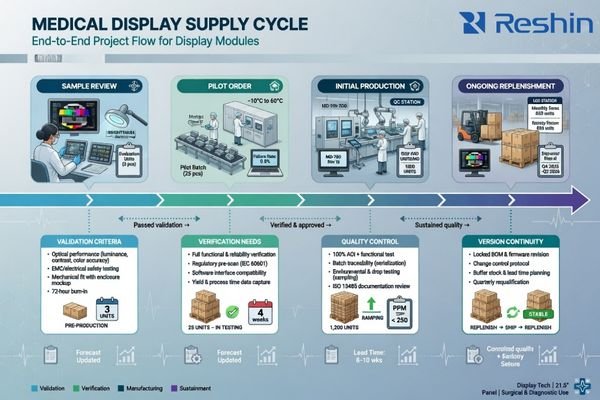
When planning a project, I always encourage buyers to think beyond the first delivery. Sample timing, pilot timing, and production timing need to fit the buyer’s internal schedule, customer commitments, and inventory planning. Repeat-order logic matters just as much. If the reorder path is unclear, the project may look fine at launch but become difficult to sustain.
Version continuity is part of the same discussion. Buyers should know how later changes are handled, how component transitions are communicated, and how a validated version is managed when the product stays in the field for longer periods. First-time buyers usually feel more confident once that supply framework is clear, because the cooperation starts to look more like a program and less like a one-off transaction.
In our discussions with first-time customers, we usually try to move the conversation from “What is the first batch lead time?” to “How will this project be supplied six months from now?” That shift matters. It helps both sides think in terms of continuity, not just first-shipment convenience.
Confirm Communication Quality and Project Support, Not Just Product Availability
When a buyer works with a supplier in another country for the first time, communication becomes part of the product experience. Good hardware cannot fully compensate for unclear technical dialogue, slow issue handling, or vague answers during evaluation.
Buyers should assess whether the supplier can understand technical requirements accurately, respond with clear analysis, and support sample review, documentation follow-up, and issue closure in a structured way. Strong communication reduces misunderstanding, rework, and internal coordination cost.

In a first cooperation, I pay close attention to how technical discussions unfold. When a partner describes a problem, can we trace it back to the real cause? When feedback comes in on a sample, can we respond with a specific explanation and a clear next step? When a document is requested, can we confirm exactly what is available and what needs follow-up? Those details tell me a lot about whether the supplier can support the project once it moves beyond the inquiry stage.
For first-time buyers, communication quality shows up in the rhythm and clarity of the responses. A mature supplier usually answers in a way that is specific, contextual, and traceable rather than vague or reactive.
In our own cooperation with OEM and distributor partners, the strongest projects are usually not the ones with the fewest questions. They are the ones where questions are answered clearly, actions are tracked, and both sides know what is being confirmed at each step. That kind of support often matters more than people expect when they begin sourcing for the first time.
FAQ About Buying a Medical Display from China for the First Time
What is the first thing buyers should send to a Chinese supplier?
The best starting point is a short project brief that includes the application, system type, signal input, target size, resolution expectation, mounting method, expected quantity, and target market. The clearer the input, the easier it is for the supplier to recommend a suitable path instead of replying with a generic quotation.
Should first-time buyers compare price first?
Price matters, but it should not be the only thing checked at the beginning. Application fit, sample-to-production alignment, documentation quality, customization scope, and supply continuity usually have a greater impact on whether the project runs smoothly later.
How can buyers tell whether a supplier’s OEM capability is practical?
The most useful test is whether the supplier can explain the scope of change, sample timing, lead-time impact, MOQ effect, and later replenishment consistency in clear terms. If those boundaries stay vague, the OEM capability may not yet be strong enough for a stable project.
What matters more than certificates in a first-time buying decision?
More important than the certificate name is whether the document package is relevant to the actual project, whether revisions are traceable, whether the sample and production stay aligned, and whether the supplier can continue supporting review and delivery over time.
A Good First Purchase Decision Reduces Risk Before the First Shipment Even Leaves the Factory
For a buyer sourcing a medical display from China for the first time, a strong first decision is rarely about finding the lowest number on the quotation sheet. It is about confirming the right things early: the application boundary, the alignment between sample and production, the documentation package, the customization scope, the supply path, and the communication process behind the product.
That early discipline makes the rest of the project easier to control. Sample validation becomes more meaningful. Internal approval moves faster. Customer delivery becomes more predictable. Repeat orders become easier to manage. If you are preparing a new medical display program, the most useful next step is to discuss it with a medical display manufacturer while the project inputs are still being defined, not after the risky assumptions have already been made.
✉️ info@reshinmonitors.com
🌐 Medical Display Manufacturer
-
Understanding these practices can help ensure smoother sourcing and better supplier relationships. ↩
-
Exploring this resource will provide insights into effective strategies for managing risks in first-time sourcing. ↩
-
Understanding the specifications and applications of a 24-inch medical monitor can help ensure you choose the right display for your needs. ↩
-
Understanding sample approval is crucial for ensuring project success and avoiding costly mistakes. ↩
-
Exploring the importance of strong documentation support can enhance your supplier collaboration and project outcomes. ↩