Many teams judge supply continuity by a simple question: can the supplier still ship the same model two or three years later? In real medical display programs, that is only part of the answer. From the way I evaluate long-cycle projects, the more useful questions are whether the baseline stays clear, whether revisions stay controlled, whether replenishment remains consistent, and whether the replacement path is already thought through before a disruption happens.
Long-term supply continuity for a medical display program is not just about later availability. It is about whether configuration, revision control, replenishment behavior, and replacement planning remain predictable enough to keep the program stable over time. A capable medical display manufacturer should be able to support that full continuity logic, not simply keep a model name active in the catalog.
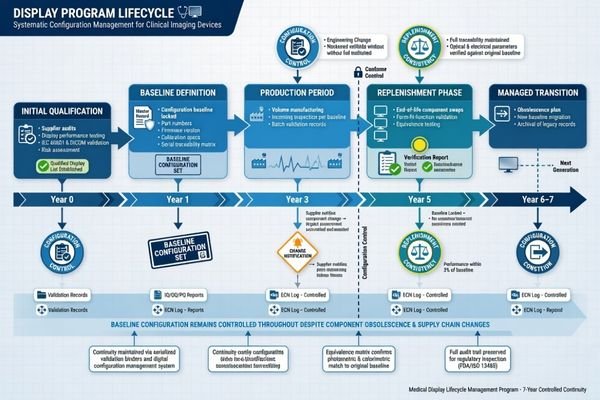
When I review long-term continuity, I do not stop at asking how many years a model may remain orderable. That is only the first layer. What I really want to know is whether future deliveries will stay aligned in configuration, behavior, documentation, and replacement logic. A supplier may still be able to ship a product under the same model name, but if the internal configuration has drifted, if firmware behavior has shifted, or if the document set is no longer aligned, the continuity of the program starts to weaken1.
For a program owner, continuity is really about keeping the program in a controlled state. That is why I pay more attention to baseline definition, change discipline, replenishment consistency, and successor planning than to availability alone. In practice, continuity is strongest when later supply still fits the original program logic instead of forcing the team to re-explain, re-check, or rework decisions that were already settled earlier.
Long-Term Supply Continuity Is Not About Shipping Alone
It is common to treat supply continuity as a simple availability question. In medical display programs, that view is usually too narrow.
Supply continuity is not only about whether a supplier can still ship later. It is about whether later batches remain controllable in configuration, behavior, and documentation, so that the program itself stays stable.
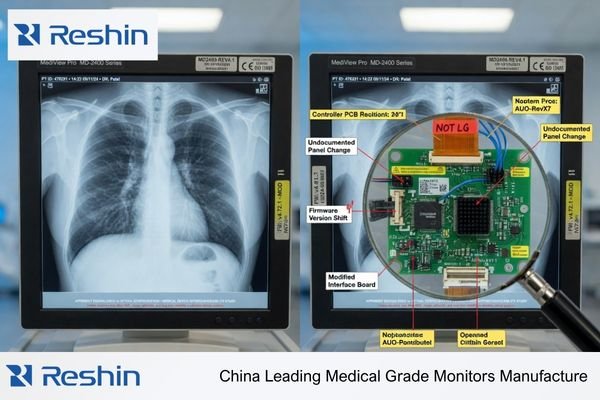
In project work, I have seen programs become difficult even when the supplier kept saying the product was still available. The real problem was that the later unit was no longer fully aligned with the earlier validated version. Sometimes the difference was small: a panel source change, a different controller behavior, a quiet firmware update, or a document package that no longer matched the shipped hardware. None of those changes sounds dramatic on its own, but together they can reduce the value of earlier validation work and create extra cost for the buyer.
That is why I do not treat continuity as a reorder question. I treat it as a program control question. The supplier may still be shipping, but if the delivered product is slowly drifting away from the approved baseline, the program is not really continuous in the way a medical project needs it to be2.
Baseline Definition and Configuration Control Come First
From an engineering standpoint, the first real control point in a long-term supply program is a clearly defined baseline. Without that, continuity becomes difficult to verify later.
Before a buyer worries about future price or future lead time, the more important step is to confirm that the validated baseline is clear and controlled. That baseline needs to cover the practical boundaries of the product, not just the model number.
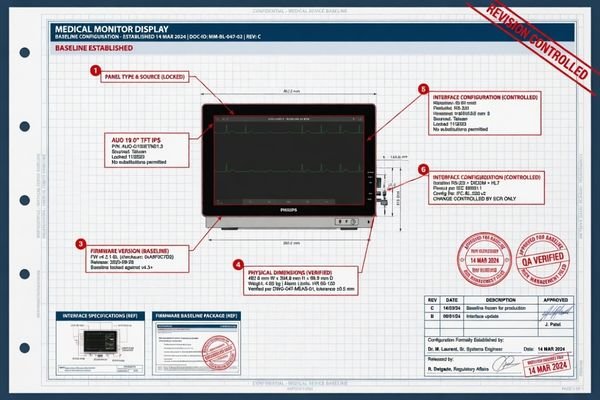
When I start a long-cycle program with a partner, I want to know exactly what the approved version of the display is. That means more than naming the model. I want the project to be clear about interfaces, dimensions, mounting logic, key operating behavior, firmware status, and the package actually being delivered with the unit. If those boundaries are vague, continuity becomes much harder to judge later because nobody is comparing future supply to a fully defined reference.
What a Practical Baseline Should Include
A useful baseline usually covers the points that can affect later deployment, replacement, or qualification work. In my experience, that includes the physical footprint of the product, the relevant interface layout, the approved firmware condition, the expected default behavior, and the accessory scope that travels with the unit. If the product is being integrated into a regulated or structured project, that clarity matters even more because later supply needs to stay aligned with something that was genuinely defined, not loosely assumed.
Why Configuration Control Matters Early
If the baseline is not properly controlled, the phrase “same model3” starts to lose value. A supplier may continue shipping under the same product name, but the buyer has no clean way to confirm whether the later units are still aligned with the earlier approved version. That is why I usually treat configuration control as the first practical sign that a continuity discussion is serious.
Change Notification and Revision Discipline Matter More Than Verbal Promises
In any multi-year medical display program, change is possible. Components move, sourcing pressure changes, and supply chains do not stay frozen forever. The more useful question is how change is handled when it happens.
A supplier’s continuity capability is usually better judged by revision discipline and change-notification logic than by verbal promises that nothing will change. What matters is whether changes are governed, communicated, and evaluated before they create avoidable program risk.
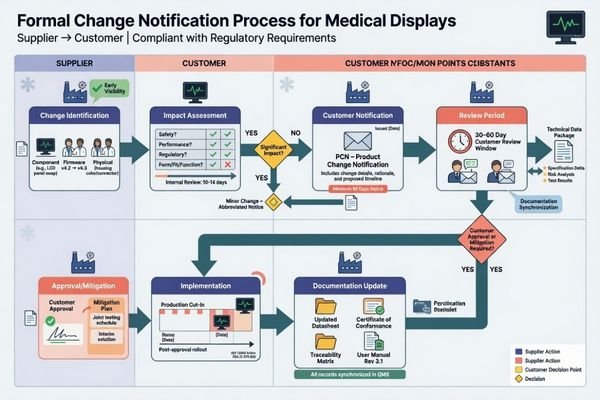
When I review a supplier’s continuity process, I look for something concrete. Is there a revision history? Is there a formal trigger for issuing a change notice? Are the technical files updated alongside the change? Can the buyer review likely impact before the next shipment goes out? Those are the kinds of signs that tell me whether the supplier understands continuity as a managed process rather than a casual promise.
This is also where a page like long-term supply and model consistency planning becomes highly relevant. The strongest long-cycle projects usually have a visible method for defining baseline, controlling revisions, and preparing for future supply conditions before a shortage or replacement event forces the issue. In practice, change itself is not always the problem. Unclear change is the problem.
Replenishment Consistency and Multi-Batch Stability Decide Real Program Continuity
The first batch rarely tells the whole story. Real continuity shows up in the second, third, and later orders, especially when those orders are tied to multi-site rollout, replacement planning, or service stock.
A program shows real continuity when later batches do not disturb the existing system logic. Replenishment consistency matters because even small differences in later supply can create extra work across installation, service, and quality management.

In long-cycle programs, I pay a lot of attention to how later shipments behave compared with the first approved batch. If later units arrive with different interface details, different default settings, slightly shifted physical dimensions, or a changed user behavior that nobody documented clearly, the field team now has to manage two realities at once. That makes deployment harder and service less tidy. It can also create internal confusion because the purchasing side may believe the program stayed constant while the engineering side knows it did not.
That is why multi-batch stability matters so much4. What I want to see is not just that batch three can ship, but that batch three still fits the logic established by batch one. When that happens, replenishment stays quiet in the best sense of the word: it supports the program without forcing the program to reorganize itself.
A Good Replacement Strategy Is Part of a Stable Program
Replacement planning is not something I leave to the end of the conversation. In a mature medical display program, it belongs inside the continuity discussion from the beginning.
A stable continuity plan should already account for successor logic, service replacement, and the possibility of controlled model transition. Replacement strategy is part of continuity, not something separate from it.
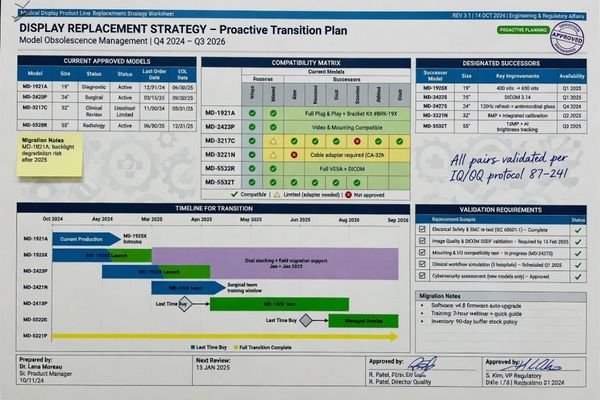
A supplier does not need to claim that the original configuration will remain unchanged forever. What I care about more is whether there is a sensible path forward if circumstances change. If a component approaches end-of-life, is there a defined successor path? If service replacements are needed, can the buyer understand what remains aligned and what needs review? If the program expands later, can future supply still be planned without guessing?
The table below is closer to the way I actually review continuity planning in a real program.
| Continuity Control Area | What Buyers Should Confirm | Why It Matters Later |
|---|---|---|
| Baseline Definition | Is the approved configuration clearly documented beyond the model name? | Later supply can only be judged against a reference that is actually defined. |
| Revision Control | Is there a usable revision history and a formal change-notification trigger? | Buyers need visibility before small changes become field-level problems. |
| Replenishment Consistency | Are later batches expected to stay aligned in behavior, interfaces, and documentation? | Multi-batch stability keeps deployment and service logic intact. |
| Replacement Strategy | Is there a clear successor or service-replacement path if the original configuration changes? | Programs need a controlled path forward, not last-minute improvisation. |
| Document Synchronization | Do technical files, approval references, and shipping reality stay aligned through change? | Documentation drift can create just as much risk as product drift. |
In my experience, this is where a lot of continuity discussions become more practical. Once the supplier can explain the replacement path, not just the current stock status, the buyer starts to see whether the program can remain manageable over time.
FAQ About Long-Term Supply Continuity for Medical Display Programs
If a model is still available, does that automatically mean supply continuity is good?
Not necessarily. A model may still be available while later supply has already shifted in configuration, behavior, or documentation. I usually judge continuity by controlled alignment, not by simple availability alone.
What should buyers confirm first when evaluating long-term continuity?
I normally start with three things: whether the baseline is clearly defined, whether revision control and change notification are real and usable, and whether replenishment plus replacement planning are already thought through.
Why is change notification so important in medical display programs?
Because medical programs often depend on earlier validation, qualification, installation logic, or internal approval history. If later changes are not visible in time, earlier work can lose part of its value and the coordination burden rises quickly.
Is replacement planning only for after-sales service?
No. A good continuity review treats replacement planning as part of early program planning. It is much better to understand the future path before a supply interruption forces urgent decisions.
A Stable Medical Display Program Depends on Controlled Continuity, Not Simple Availability
If I reduce the whole topic to one practical conclusion, it is this: the real question is not whether the supplier can still ship later, but whether the program can still run in a controlled state later. That is a much better test of long-term continuity.
In my own project work, I usually give the most weight to baseline clarity, configuration control, change notification, replenishment consistency, and replacement planning. Those are the areas that sit closest to the real risks carried by program owners, procurement teams, engineering teams, and long-cycle deployment plans. If you are reviewing a multi-year medical display program, the most useful next step is usually to confirm your baseline, revision path, replenishment logic, and successor strategy before locking future supply with a medical display manufacturer.
✉️ info@reshinmonitors.com
🌐 Medical Display Manufacturer
-
"Configuration lifecycle management maturity model – ScienceDirect", https://www.sciencedirect.com/science/article/abs/pii/S0166361518301301. Studies in configuration management show that drift in system configuration, firmware behavior changes, and misaligned documentation increase integration errors and support overhead, thereby weakening overall program continuity. Evidence role: mechanism; source type: paper. Supports: if the internal configuration has drifted, if firmware behavior has shifted, or if the document set is no longer aligned, the continuity of the program starts to weaken.. Scope note: Most studies focus on software-intensive systems; effects may differ in purely hardware contexts. ↩
-
"21 CFR Part 820 — Quality Management System Regulation – eCFR", https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820. FDA Design Control Guidance and 21 CFR 820.30 require that medical device design changes be thoroughly documented and controlled to preserve continuity of the approved design history. Evidence role: expert_consensus; source type: government. Supports: medical projects require strict baseline continuity through formal change control. Scope note: Applies to U.S. FDA–regulated devices; international regulatory requirements may differ. ↩
-
"Configuration Management – Defense Acquisition University", https://content1.dau.edu/DAUMIG_se-brainbook_189/content/Management%20Processes/Configuration-Management.html. ISO 10007 outlines that without a properly controlled baseline in configuration management, product identifiers such as model numbers cannot guarantee consistency across deliveries. Evidence role: expert_consensus; source type: institution. Supports: If the baseline is not properly controlled, the phrase “same model” starts to lose value.. Scope note: The standard addresses configuration management broadly and may not explicitly use the phrase “same model.” ↩
-
"Continuous Manufacturing vs Batch Manufacturing | GE Vernova", https://www.gevernova.com/software/blog/continuous-batch-manufacturing-differences. Lean manufacturing principles, as described in the Toyota Production System article on Wikipedia, emphasize the importance of maintaining consistent production batches to reduce waste and ensure smooth replenishment without process reorganization. Evidence role: mechanism; source type: encyclopedia. Supports: Maintaining multi-batch stability is essential because it ensures that subsequent production runs adhere to the logic and specifications of the initial batch, thereby preventing supply chain disruptions and program reorganizations.. Scope note: Focuses on lean manufacturing contexts and may not directly address specific long-cycle program scenarios. ↩



