When buyers evaluate a medical display supplier, documentation review is usually part of the decision process from the very beginning. These requests are not simply about collecting files. They are used to reduce uncertainty, compare suppliers more objectively, and confirm whether a proposed product and supplier can support a real medical device project.
Buyers usually request documentation from a medical display supplier to verify supplier legitimacy, quality-system maturity, product definition, market-planning readiness, and long-term project control. Different document sets support different decisions, which is why document requests often change from supplier screening to RFQ, sample validation, registration planning, and shipment preparation.

A supplier’s documentation package often reveals more than the files themselves. Clear, structured, and timely documents usually indicate better internal coordination, stronger process control, and a more reliable support path for customization, validation, registration, and supply continuity. By contrast, unclear or inconsistent documentation can increase project risk even before technical evaluation is complete.
Quick Guide: What Buyers Usually Request and Why
Before going into detail, it helps to map the most common document groups to the decisions they support.
| Document Group | What Buyers Use It to Verify | Typical Project Stage | If Missing, What Risk Increases |
|---|---|---|---|
| Company-Level Documents | Supplier identity, manufacturing role, business foundation, quality-system maturity | Initial supplier screening | Supplier credibility risk |
| Product Documents | Product definition, interface fit, mechanical fit, technical clarity | RFQ and technical review | Integration and quotation risk |
| Compliance & Registration Documents1 | Market-path support, labeling readiness, applicable regulatory alignment | Market planning, registration preparation | Market-entry and approval risk |
| Project-Control Documents | Validation discipline, revision control, change handling, traceability support | Sample evaluation, pilot, long-term cooperation | Lifecycle control risk |
| Stage-Based Document Packages | Whether the right files are available at the right time | Across the full project cycle | Delays, rework, and decision gaps |
This structure matters because buyers rarely request every file at once. In most cases, they request different document sets at different points to support a specific decision.
Why Buyers Request Documentation in the First Place
Documentation requests are usually tied to practical decision-making rather than formal process alone. Buyers need evidence that the supplier is real, the product is sufficiently defined, the project can move forward on a stable basis, and the intended market path is supportable.
The purpose of documentation review is usually risk reduction. Buyers use documents to confirm who the supplier is, what the product actually is, how clearly the project is defined, and whether the supplier can support the program beyond the first quotation or sample.
A useful way to understand documentation requests is to view them as answers to different buyer questions:
- Is this supplier a real manufacturer or only a trading intermediary?
- Is the product defined clearly enough for technical review2?
- Can this supplier support the intended market path?
- Are changes, revisions, and project records handled in a controlled way?
- Will documentation remain aligned with the product during later project stages?
These questions explain why documentation is often reviewed alongside quotations, samples, drawings, and technical discussions rather than as a separate administrative step.
Company-Level Documents Buyers Usually Ask for First
At the beginning of supplier evaluation, buyers usually start with company-level documents. These files are used to establish a basic level of trust before technical review goes deeper.
Company-level documents are usually requested first because buyers need to verify supplier identity, manufacturing role, quality-system foundation, and general business credibility before investing time in detailed technical evaluation.

Verifying Supplier Identity and Manufacturing Role
A company profile, factory overview, and basic business credentials help buyers understand whether the supplier is a real manufacturer, what its operating scope looks like, and whether the business relationship can be verified on a practical level.
These files often help answer questions such as:
- Is the supplier actually involved in manufacturing?
- Does the company have a stable operational base?
- Is the supplier’s experience relevant to medical display projects?
- Does the supplier appear structured enough for longer-term cooperation?
For buyers, this is the first filter. If the business foundation is unclear, later technical review becomes much less meaningful.
Confirming Quality-System Maturity3
Quality-system documents are another early-stage requirement, especially when the buyer is evaluating suppliers for regulated or long-lifecycle medical projects. In many cases, buyers want to see whether the supplier works under a recognized quality framework and whether internal control appears structured rather than informal.
A clear quality-system package usually supports confidence in:
- process consistency
- document control
- production discipline
- traceability expectations
- readiness for structured project cooperation
This is also where buyers begin to judge whether a supplier is likely to be manageable during validation, design changes, and later production stages.
Product Documents Used for Technical Review and Supplier Evaluation
Once the supplier has passed basic credibility review, attention usually shifts to the product itself. At this stage, buyers need technical documentation that is detailed enough to support RFQ, engineering review, and internal discussion.
Product documents are used to determine whether the display definition is clear, whether the proposed product can fit the target system, and whether the technical assumptions are stable enough for quotation, sampling, and integration planning.

Documents That Support Technical Fit Assessment
The most commonly requested product-level files usually include:
- datasheets
- specification sheets
- interface summaries
- mechanical drawings
- user manuals or draft operating documents
- key performance and test-related information
These documents help buyers evaluate whether the product is defined clearly enough for internal technical review. They also help answer practical questions such as:
- Can the display fit the intended enclosure or workstation layout?
- Is the interface behavior documented clearly enough for system review?
- Does the performance definition match the use case?
- Is the product definition stable enough for quotation and sample planning?
Why Product Clarity Matters Before Sampling
A physical sample is useful, but a sample without clear technical documentation4 often creates more ambiguity instead of reducing it. Buyers usually want the product definition to be sufficiently documented before sampling begins so that engineering discussions, quotation assumptions, and internal approvals are built on the same reference.
When product documentation is incomplete, common problems include:
- unclear RFQ scope
- mismatched interface expectations
- uncertainty around mounting and enclosure planning
- confusion between standard features and custom requirements
- avoidable rework during sample review
For this reason, product documents are often one of the most heavily reviewed parts of the supplier package.
Compliance and Registration Documents Buyers Request for Market Planning
Technical suitability alone is not enough for most medical device projects. Buyers also need to understand whether the proposed display can be supported along the intended market path and whether the supplier can cooperate on compliance-related documentation when needed.
Compliance and registration-related document requests are usually tied to market planning. Buyers want to know whether the supplier can support required declarations, applicable standards information, labeling details, and other materials needed for evaluation, registration preparation, or customer review.

Documents Buyers Often Review for Market Readiness
Depending on the market and project stage, buyers may request documents such as:
- declarations of conformity
- certificate copies
- applicable standards information
- labeling details
- product identification information
- market-specific support documents
- technical file support references5, where relevant
The purpose is usually not to collect documents for their own sake, but to confirm whether the supplier can support the project beyond the engineering stage.
Why This Section Is Often About Support Capability
In practice, many buyers are not only evaluating the document set itself. They are also evaluating whether the supplier can provide structured compliance and registration support during the project. A supplier that can explain document scope clearly, maintain revision consistency, and cooperate on technical file preparation usually presents lower project risk than one that can only provide scattered certificates without context.
This is especially important when the buyer is planning for:
- multi-country rollout
- customer audit requirements
- registration preparation
- first-shipment documentation alignment
- document maintenance after initial approval
Project Documents That Reflect Real Cooperation Capability
Company documents and product documents help buyers understand who the supplier is and what the product is. Project documents go one step further. They show how the supplier manages the product over time.
Project-level documentation is often one of the clearest indicators of real cooperation capability because it reflects how the supplier handles validation, revisions, controlled changes, document-to-sample consistency, and long-term project support.

Documents That Show Operational Discipline
Buyers often pay close attention to project-related materials such as:
- sample validation records
- revision history
- document revision control
- change-notification logic
- traceability references
- QC checkpoint records
- document-to-sample consistency evidence
These files help buyers judge whether the supplier can support a real project environment rather than only provide a product brochure and quotation.
Why These Files Matter for Long-Lifecycle Programs
For medical device projects, the challenge is not only getting the first sample approved. The challenge is maintaining consistency as the project moves through evaluation, pilot, registration support, and ongoing supply.
Project-control documents help buyers assess whether the supplier can:
- manage changes in a controlled way
- communicate revisions clearly
- keep document versions aligned with product reality
- support repeatability across later batches
- reduce risk during longer cooperation cycles
This is often where supplier maturity6 becomes much more visible.
When Buyers Usually Request Different Documents at Different Stages
Documentation review usually follows the logic of the project itself. Buyers do not always ask for every file at the same time because each stage has a different decision focus.
Document timing matters because supplier screening, RFQ review, sample validation, registration planning, and shipment preparation do not require exactly the same evidence. The most effective document flow delivers the right files at the point where they support the next decision.
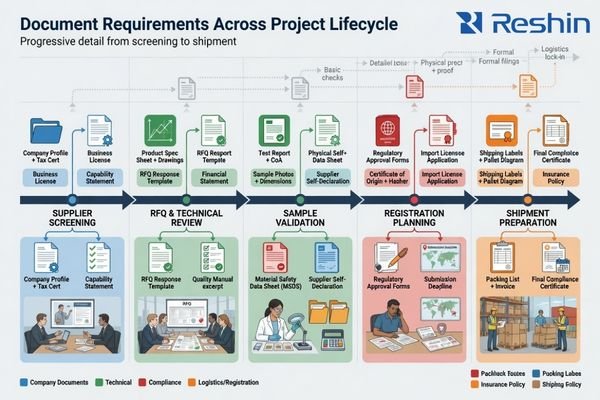
Before Supplier Screening
At this stage, buyers are usually focused on supplier legitimacy and quality-system foundation.
Typical requests include:
- company profile
- factory overview
- quality-system information
- core business credentials
The main question is whether the supplier is credible enough to move into deeper review.
Before RFQ and Technical Review
At this stage, buyers need the product to be defined clearly enough for technical evaluation and quotation alignment.
Typical requests include:
- datasheets
- mechanical drawings
- interface summaries
- product definition materials
The main question is whether the proposed product can realistically fit the project.
Before Sample Validation
Here the focus shifts from paper definition to controlled evaluation.
Typical requests include:
- sample-related records
- key test support information
- user-facing product documentation
- revision references
- validation support inputs
The main question is whether the product can be evaluated against clear assumptions.
Before Registration or First Shipment
At later stages, documentation usually becomes more structured and more market-specific.
Typical requests include:
- declarations
- labeling details
- standards-related information
- shipment-support document packages
- registration-support materials, where needed
The main question is whether the documentation set is sufficient to support downstream approval, customer requirements, or market-entry preparation.
| Project Stage | Primary Buyer Decision | Documents Commonly Requested |
|---|---|---|
| Initial Screening | Is the supplier credible enough to evaluate further? | Company profile, factory overview, quality-system information |
| RFQ & Technical Review | Is the product defined well enough for fit assessment and quotation? | Datasheet, specification sheet, interface summary, mechanical drawing |
| Sample & Validation | Can the product be evaluated in a controlled and meaningful way? | Validation-related records, user documentation, revision references |
| Registration & Shipment Preparation | Can the project move forward with the required market and customer documentation? | Declarations, labeling details, standards-related information, structured support package |
What Clear Documentation Usually Signals to the Buyer
Experienced buyers often treat documentation quality as an early signal of how the supplier is likely to perform in the wider project. Clear documentation suggests that internal coordination is stronger and that future communication may be more predictable.
Well-prepared documentation usually signals stronger internal control, better cross-team coordination, clearer revision handling, and a more reliable support structure for customization, validation, registration, and continuity planning.

In practical supplier evaluation, document readiness often shapes buyer perception in the following areas:
- communication quality
- responsiveness to document requests
- project discipline
- change-management reliability
- ability to support technical review and downstream compliance work
- confidence in long-term cooperation
For many buyers, this signal is important because document quality often appears before the supplier is tested at larger project scale. In that sense, documentation is not only evidence. It is also an indicator of operational maturity.
FAQ
What documents do buyers usually request before RFQ?
Before RFQ, buyers usually focus on two areas: supplier credibility and product definition. Common requests include company profile materials, quality-system information, datasheets, interface summaries, and mechanical drawings. The goal is to confirm that the supplier is credible and that the proposed display is defined clearly enough for technical and commercial review.
Do buyers usually need registration-related documents before sampling?
Not always. In many projects, registration-related document requests become more important after early supplier screening and technical fit review. However, if the target market is already fixed or the buyer is planning for a regulated rollout, buyers may request compliance-related materials earlier to reduce later approval risk.
Which documents best show real change-control capability?
Project-level documents usually reveal this most clearly. Revision history, document revision control, change-notification logic, sample validation records, and traceability-related references often provide stronger evidence of change-control discipline than basic product brochures or general certificates alone.
Conclusion
When buyers request documentation from a medical display supplier, they are usually making practical, risk-based decisions rather than simply collecting files. Different documents help them verify different things: supplier legitimacy, product clarity, market-planning readiness, and the supplier’s ability to support a controlled project over time.
The strongest documentation packages do more than provide information. They show that the supplier can communicate clearly, manage revisions responsibly, and support the project through technical review, validation, market preparation, and ongoing cooperation. For buyers evaluating long-lifecycle medical display projects, that signal is often just as important as the files themselves.
If documentation support is likely to affect your evaluation, registration planning, or project coordination, it helps to review what structured compliance and registration support is actually available before the project moves too far forward.
-
Exploring Compliance & Registration Documents helps ensure regulatory alignment and successful market entry, reducing approval risks. ↩
-
Exploring technical reviews can enhance your knowledge of product evaluation and supplier assessment. ↩
-
Understanding Quality-System Maturity is crucial for ensuring supplier reliability and compliance in regulated industries. ↩
-
Understanding the role of technical documentation can enhance your product development process and ensure clarity. ↩
-
Exploring technical file support references can enhance your knowledge of project documentation and compliance. ↩
-
Exploring supplier maturity can reveal insights into their capability to manage long-term projects effectively, ensuring quality and compliance. ↩



