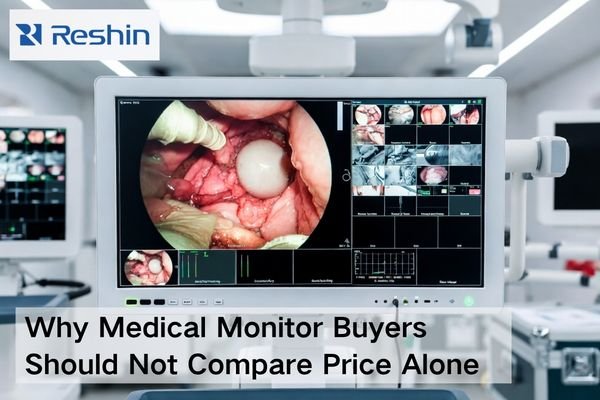
Top 7 Most Recommended Surgical Monitors for Low-Latency OR Workflows (2026)
For 2026, the most recommended surgical monitors for low-latency OR workflows must combine true 4K clarity, optimized signal processing, robust 12G-SDI/HDMI/DP connectivity, and hygiene-focused design to keep surgical video in perfect sync with real-world movement. This Top 7 list compares how leading 31–32” class 4K surgical monitors from Sony, Reshin, LG, EIZO, and FSN perform in demanding OR environments.