How Manufacturer-Side Registration Support Works in Medical Display Projects
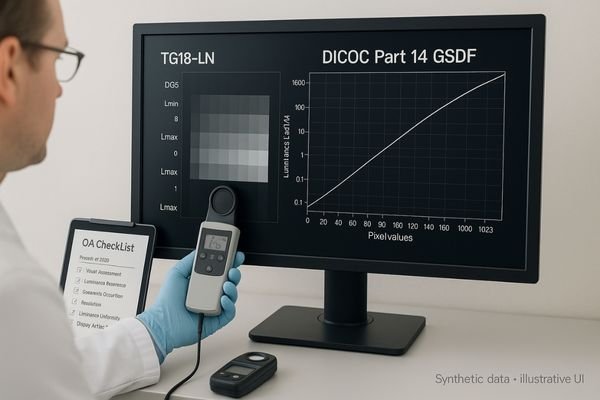
Manufacturer-side registration support in medical display projects is not about the supplier completing registration on the buyer’s behalf. It is a structured form of compliance and registration support built around requirements alignment, controlled documentation, verification cooperation, technical clarification, and disciplined change management, so the buyer’s own registration preparation can move forward with fewer blind spots and less avoidable rework.