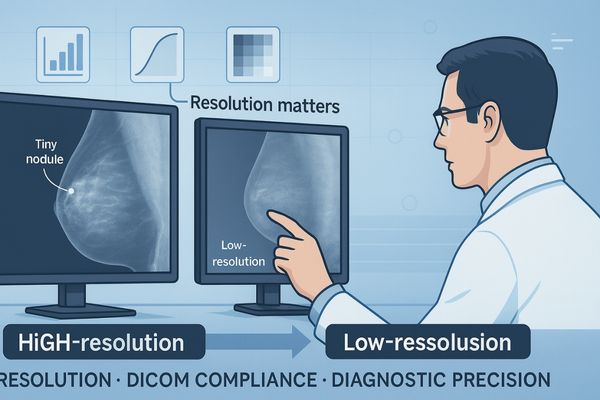
DICOM Curve Interpretation of Medical-Grade Displays
The DICOM curve is the foundation of medical-grade displays, ensuring grayscale levels match human visual sensitivity. By aligning technology with visual science, DICOM GSDF guarantees consistent image quality, enabling greater diagnostic confidence.