When Procuring Medical-Grade Monitors, What Key Clauses Should an RFP Include?
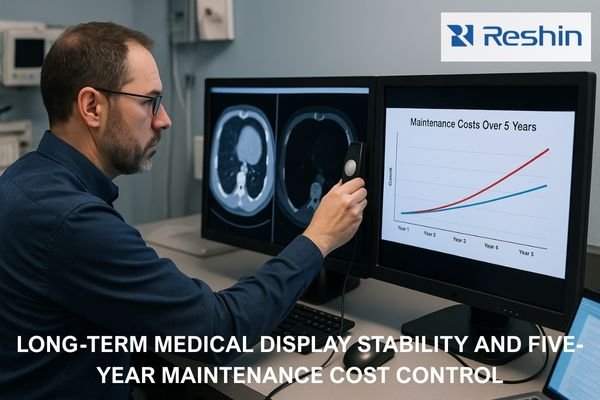
Medical-grade monitor RFPs should include enforceable clauses covering compliance traceability, measurable performance acceptance criteria, documented baseline configurations, and lifecycle controls that prevent drift after updates and replacements. Strong RFP language converts operational requirements into verifiable deliverables with defined test methods and pass/fail criteria.