How to Choose a Surgical Monitor for Endoscopy System Projects
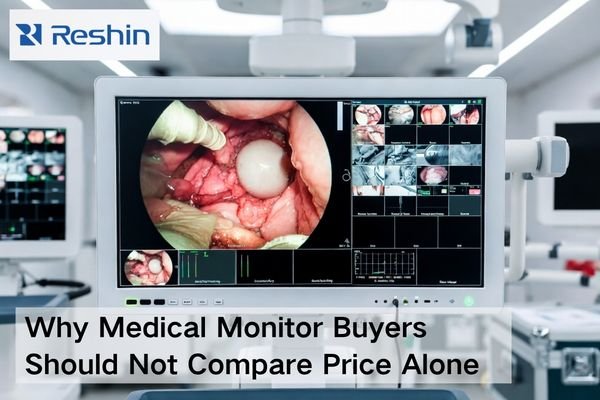
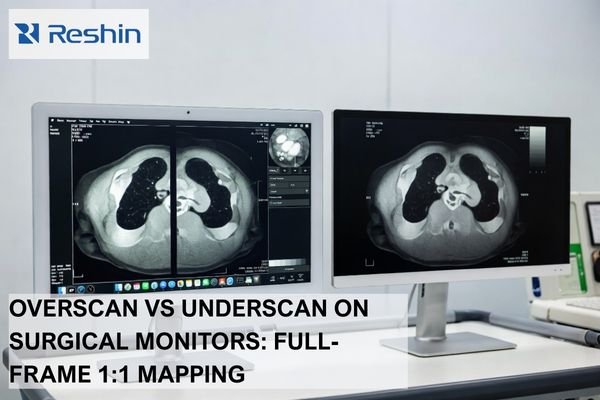
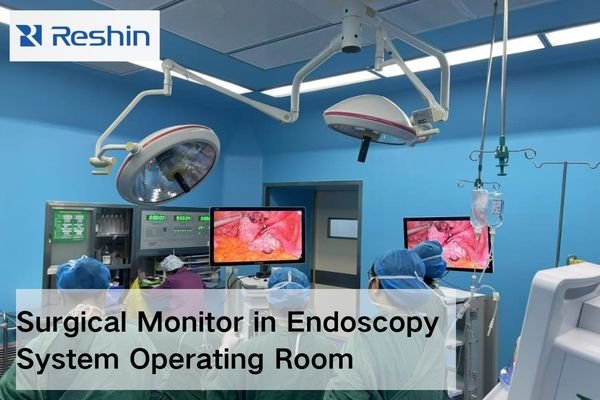
To choose a surgical monitor for an endoscopy project, buyers should start with four checks: processor output and signal path, viewing distance and mounting position, required image detail, and rollout continuity. A qualified surgical monitor manufacturer should help match the monitor to the system, not just promote a stronger standalone specification.